In modern healthcare, intravenous therapy plays a vital role in patient treatment. However, many people—including some healthcare professionals—may not fully understand the crucial distinction between blood transfusion sets and IV infusion sets. While both devices deliver fluids directly into a patient’s bloodstream, they are designed for fundamentally different purposes and should never be used interchangeably.
This comprehensive guide explores the key differences between blood transfusion sets and IV infusion sets, explaining why proper selection is essential for patient safety and optimal clinical outcomes.
What is an IV Infusion Set?
An IV infusion set (also called an intravenous administration set) is a medical device designed to deliver medications, fluids, electrolytes, and nutrients directly into a patient’s vein. These sets are the workhorse of hospital care, used countless times daily for routine medical treatments. Modern safety IV catheters have significantly improved the safety profile of intravenous access, reducing needlestick injuries and infection risks.
Common Uses of IV Infusion Sets
- Saline solutions and hydration therapy
- Dextrose and glucose administration
- Antibiotic delivery
- Pain medication infusion
- Chemotherapy drugs
- Total parenteral nutrition (TPN)
- Electrolyte replacement
IV infusion sets are versatile tools designed for a wide range of crystalloid and colloid solutions, but they lack the specialized features required for safe blood product administration.
What is a Blood Transfusion Set?
A blood transfusion set is a specialized medical device engineered specifically for the administration of blood and blood products. Unlike standard IV infusion sets, blood transfusion sets incorporate unique design features that protect blood components from damage and prevent potentially life-threatening complications. The blood collection process itself requires specialized equipment, including blood collection needles designed for safe and efficient venipuncture.
Common Uses of Blood Transfusion Sets
- Whole blood transfusion
- Packed red blood cells (PRBCs)
- Fresh frozen plasma (FFP)
- Platelet concentrates
- Cryoprecipitate
- Other blood component therapy
Blood transfusion sets are purpose-built for the unique properties of blood, which is far more complex than simple IV fluids.
Key Differences Between Blood Transfusion Sets and IV Infusion Sets
1. Filter Design and Specifications
The most critical difference between blood transfusion sets and IV infusion sets lies in their filtration systems.
Blood Transfusion Sets:
- Equipped with specialized blood filters (typically 170-260 microns)
- Designed to trap blood clots, fibrin strands, and cellular debris
- Prevent microemboli from entering the patient’s circulation
- Mandatory component for all blood product administration
- Filter prevents potential complications from aggregated cells
IV Infusion Sets:
- Feature basic drip chambers with simple mesh filters or no filter at all
- Designed only to prevent large particles from IV solutions
- Not equipped to handle the viscosity of blood products
- Cannot filter microaggregates found in stored blood
Using an IV infusion set for blood transfusion poses serious safety risks, as microclots and debris could reach vital organs, potentially causing pulmonary complications or stroke.
2. Material Composition and Tubing Design
Blood Transfusion Sets:
- Manufactured from materials that minimize hemolysis (red blood cell destruction)
- Wider internal diameter to accommodate blood viscosity
- Designed to reduce mechanical stress on blood cells
- Compatible with blood warming devices when needed
- May include Y-type connectors for saline priming
IV Infusion Sets:
- Standard medical-grade PVC or polyethylene tubing
- Narrower bore suitable for low-viscosity solutions
- Not optimized for preserving cellular blood components
- May cause hemolysis if used with blood products
The use of safety IV catheters in conjunction with appropriate infusion sets has become a standard practice to minimize complications and ensure optimal patient outcomes.
3. Flow Rate Capabilities
Blood transfusion sets and IV infusion sets differ significantly in their flow rate characteristics:
Blood Transfusion Sets:
- Designed for controlled blood flow rates
- Typically deliver 10-15 drops per milliliter
- Accommodate the higher viscosity of blood products
- Support both standard and rapid transfusion protocols
IV Infusion Sets:
- Optimized for crystalloid and colloid solutions
- Usually deliver 15-20 drops per milliliter (macro-drip) or 60 drops per milliliter (micro-drip)
- Not suitable for blood’s thicker consistency
4. Safety Features and Quality Standards
Blood Transfusion Sets:
- Must meet stringent FDA and international blood banking standards
- Undergo rigorous biocompatibility testing
- Include additional safety features like air vents and injection ports
- Sterile and pyrogen-free certification required
- Often include built-in saline flush capability
IV Infusion Sets:
- Meet general medical device standards
- Designed for broader range of solutions
- May lack specific blood compatibility testing
Clinical Applications: When to Use Each Type
Scenarios Requiring Blood Transfusion Sets
Blood transfusion sets must be used for:
- Emergency hemorrhage management – Trauma patients requiring rapid blood replacement
- Surgical procedures – Anticipated or ongoing blood loss during operations
- Anemia treatment – Patients with severe anemia requiring packed red blood cells
- Coagulation disorders – Administration of clotting factors and platelets
- Cancer treatment – Patients with treatment-related blood component deficiencies
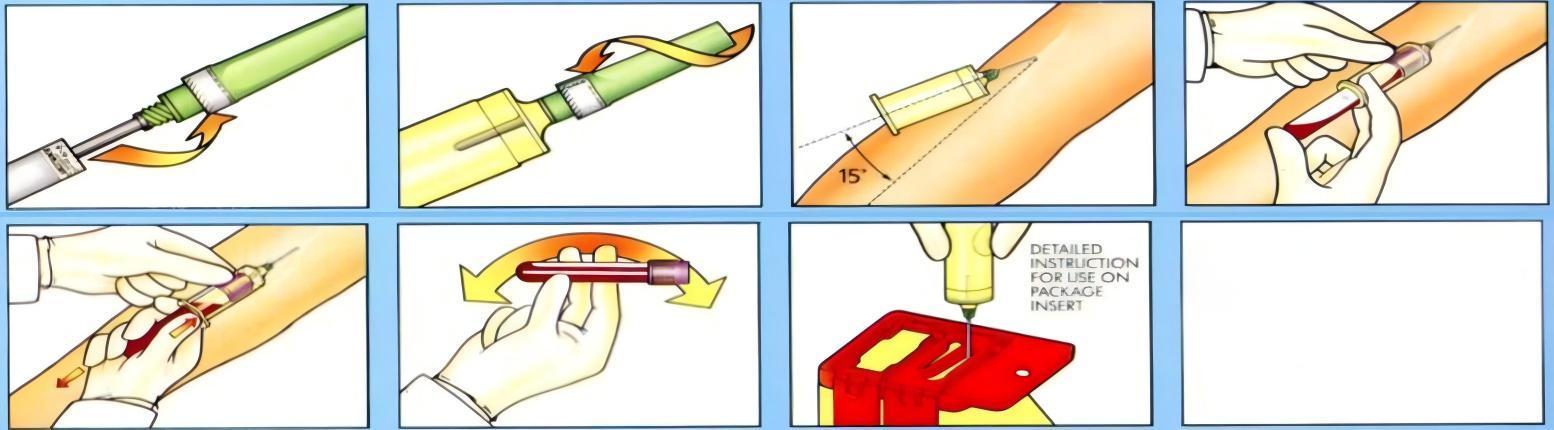
Before any blood transfusion procedure, proper blood collection using appropriate blood collection needles is essential to ensure sample integrity for cross-matching and compatibility testing.
Scenarios Requiring IV Infusion Sets
IV infusion sets are appropriate for:
- Hydration therapy – Dehydration treatment with saline or lactated Ringer’s
- Medication administration – Antibiotics, antivirals, and other drugs
- Nutritional support – TPN and lipid emulsions
- Maintenance fluids – Post-operative or chronic care fluid management
- Chemotherapy – Cancer treatment drug delivery
Why Blood Transfusion Sets and IV Infusion Sets Cannot Be Interchanged
Risks of Using IV Infusion Sets for Blood Transfusion
Using an IV infusion set instead of a blood transfusion set can result in:
- Microembolism – Unfiltered blood clots entering circulation
- Hemolysis – Destruction of red blood cells, releasing harmful substances
- Transfusion reactions – Increased risk of adverse events
- Reduced efficacy – Damaged blood components provide less therapeutic benefit
- Patient harm – Potential organ damage or death in severe cases
Drawbacks of Using Blood Transfusion Sets for IV Fluids
While less dangerous than the reverse, using blood transfusion sets for regular IV infusion is:
- Cost-ineffective – Blood transfusion sets are more expensive
- Wasteful – Specialized equipment used unnecessarily
- Flow rate issues – May not provide optimal flow for certain IV medications
Best Practices for Healthcare Professionals
Selection Guidelines
- Always verify the intended infusion – Blood products require blood transfusion sets
- Check facility protocols – Follow institutional policies on set selection
- Inspect equipment – Ensure blood transfusion sets have intact filters
- Educate team members – Ensure all staff understand the critical differences
- Double-check before connecting – Verify correct set type before initiating infusion
When establishing intravenous access, healthcare professionals should use safety IV catheters to minimize occupational exposure risks while ensuring secure vascular access for the appropriate infusion set.
Quality Assurance
- Implement color-coding systems to differentiate blood transfusion sets from IV infusion sets
- Conduct regular staff training on proper equipment selection
- Maintain clear labeling and storage separation
- Document all transfusions with equipment specifications
Common Misconceptions About Blood Transfusion Sets and IV Infusion Sets
Myth 1: “Any IV tubing can be used for blood transfusion in emergencies”
Reality: Even in emergency situations, blood transfusion sets must be used. The filter in blood transfusion sets is essential for patient safety, regardless of urgency.
Myth 2: “The difference is just the filter; I can add a filter to an IV set”
Reality: Blood transfusion sets have multiple design differences beyond filtration, including material composition, tubing diameter, and flow characteristics specifically engineered for blood products.
Myth 3: “Small volume transfusions don’t need special sets”
Reality: All blood products, regardless of volume, require blood transfusion sets. Even a single unit of packed red blood cells must be administered through appropriate equipment.
Myth 4: “Blood transfusion sets and IV infusion sets from the same manufacturer are interchangeable”
Reality: Despite sharing a manufacturer, these devices are distinct products designed for different clinical applications and cannot substitute for one another.
Regulatory and Standard Requirements
Both blood transfusion sets and IV infusion sets must comply with healthcare regulations:
United States Standards
- FDA 21 CFR Part 800-1299 medical device regulations
- AABB (American Association of Blood Banks) standards for blood transfusion sets
- ISO 1135-4 standards for transfusion equipment
- USP Chapter <797> sterility requirements
International Standards
- ISO 8536 series for infusion equipment
- European CE marking requirements
- WHO guidelines for blood transfusion safety
Healthcare facilities must ensure that both blood transfusion sets and IV infusion sets meet all applicable standards and undergo regular quality verification.
The Future of Transfusion and Infusion Technology
Medical device manufacturers continue to innovate in both blood transfusion sets and IV infusion sets:
Emerging Developments
- Smart infusion systems – Integration with electronic health records
- Advanced filtration – Improved filter designs for enhanced safety
- Closed system technologies – Reduced contamination risk
- Needleless connectors – Better infection control
- RFID tracking – Enhanced inventory management and traceability
These innovations aim to improve patient outcomes while maintaining the fundamental distinction between blood transfusion sets and IV infusion sets. The evolution of safety IV catheters and blood collection needles demonstrates the medical industry’s commitment to continuous safety improvements across all aspects of vascular access and fluid administration.
Conclusion: Understanding the Critical Distinction
The difference between blood transfusion sets and IV infusion sets is not merely technical—it’s a matter of patient safety. While both devices deliver fluids intravenously, blood transfusion sets are specifically engineered with specialized filters, biocompatible materials, and design features that protect blood components and prevent serious complications.
Key takeaways:
- Blood transfusion sets must always be used for blood and blood products
- IV infusion sets are designed for medications, hydration, and nutrition
- The two types of sets are never interchangeable
- Proper selection prevents hemolysis, microembolism, and transfusion reactions
- Healthcare professionals must understand these differences to ensure patient safety
By recognizing the critical distinctions between blood transfusion sets and IV infusion sets, healthcare providers can make informed equipment selections that protect patients and optimize therapeutic outcomes. Whether administering life-saving blood products or routine IV medications, using the correct infusion equipment is a fundamental aspect of safe, high-quality patient care.
From the initial blood draw using appropriate blood collection needles to the final administration through proper infusion sets and safety IV catheters, every step in the transfusion and infusion process demands attention to detail and adherence to established safety protocols.
Medical Disclaimer: This article is for informational purposes only and should not replace professional medical training or institutional protocols. Always follow your facility’s guidelines and consult with qualified healthcare professionals regarding patient care decisions.