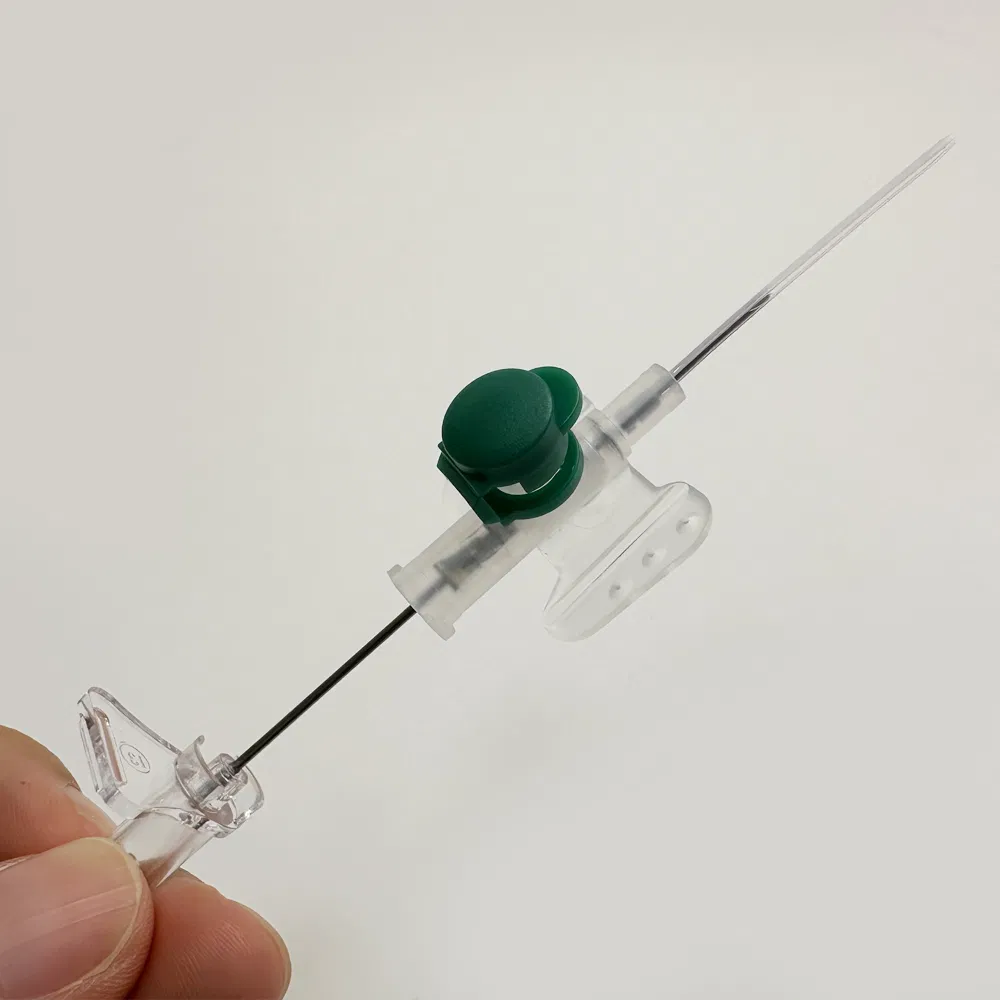
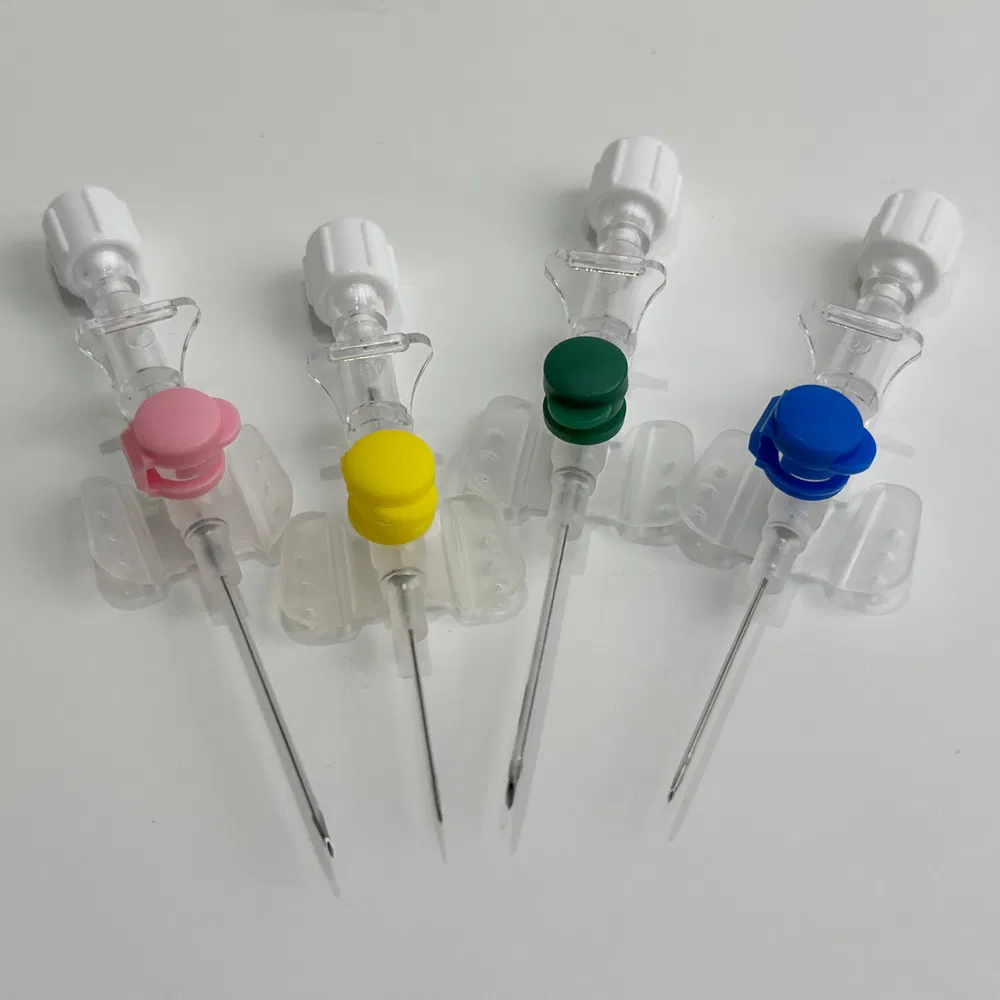
Selecting the right Safety IV Cannula manufacturer is a critical decision that impacts patient safety, regulatory compliance, and your bottom line. With numerous suppliers offering Winged IV Catheter and Hypoallergenic IV Cannula products, understanding the key differences between manufacturers helps procurement professionals make informed decisions that support long-term business success.
This comprehensive guide reveals what distinguishes quality manufacturers from mediocre suppliers and provides a strategic framework for building lasting partnerships in the medical device supply chain.
Part 1: Critical Differences Between Safety IV Cannula Manufacturers
1.1 Manufacturing Standards & Quality Control
Top-Tier Manufacturers:
- Maintain ISO 13485 certified facilities with validated cleanroom environments
- Implement automated quality inspection systems for every Safety IV Cannula unit
- Conduct batch testing including biocompatibility, sterility, and flow rate verification
- Provide full traceability from raw materials to finished Hypoallergenic IV Cannula products
- Offer comprehensive Certificate of Analysis (CoA) with each shipment
Lower-Tier Suppliers:
- May lack proper certification or operate under expired credentials
- Rely on spot-check sampling rather than systematic quality control
- Cannot provide detailed manufacturing documentation
- Offer limited batch traceability for Winged IV Catheter products
Buyer Impact: Quality control gaps lead to higher defect rates, increased patient complications, and potential regulatory violations that can halt your operations.
1.2 Material Selection & Biocompatibility
Premium Manufacturers:
- Source medical-grade polymers from certified suppliers (FEP, PU, silicone)
- Ensure all Hypoallergenic IV Cannula materials are latex-free and DEHP-free
- Conduct USP Class VI biocompatibility testing on all patient-contact materials
- Use ultra-sharp needle technology that minimizes insertion trauma
- Apply hydrophilic coatings to reduce friction and phlebitis risk
Standard Manufacturers:
- May use generic materials without comprehensive biocompatibility documentation
- Limited material options for Safety IV Cannula customization
- Basic needle sharpness without advanced grinding technology
- Inconsistent catheter material quality across production batches
Buyer Impact: Material quality directly affects patient outcomes, allergic reaction rates, and catheter dwell time—key performance indicators for healthcare facilities.
1.3 Design Innovation & Engineering
Innovative Manufacturers:
- Invest in R&D for next-generation Winged IV Catheter designs
- Offer passive safety mechanisms that activate automatically
- Develop ergonomic wing configurations based on clinical feedback
- Integrate blood control technology to minimize exposure risk
- Design self-sealing ports with leak-proof valve systems
Conventional Manufacturers:
- Produce basic Safety IV Cannula designs without innovation
- Rely on outdated safety mechanisms requiring manual activation
- Standard wing designs without ergonomic optimization
- Limited customization capabilities
Buyer Impact: Advanced designs improve clinical outcomes, enhance healthcare worker safety, and differentiate your product offerings in competitive markets.
1.4 Regulatory Compliance & Documentation
Reputable Manufacturers:
- Hold current FDA 510(k) clearance and CE marking under MDR
- Maintain active registrations in target markets (CFDA, PMDA, TGA)
- Provide complete technical files and Declaration of Conformity
- Offer regulatory support for Hypoallergenic IV Cannula market entry
- Conduct post-market surveillance and vigilance reporting
Questionable Suppliers:
- Claim certifications without providing verifiable documentation
- Offer generic certificates that don’t match specific Winged IV Catheter models
- Cannot support regulatory submissions in regulated markets
- Lack systematic adverse event monitoring
Buyer Impact: Regulatory non-compliance exposes you to legal liability, product recalls, and market access barriers that can destroy your business reputation.
1.5 Customization Capabilities
Full-Service Manufacturers:
- Offer comprehensive OEM/ODM for Safety IV Cannula specifications
- Customize gauge sizes, catheter lengths, and wing configurations
- Provide private labeling with your branding on hubs and packaging
- Develop custom color-coding systems for easy identification
- Create market-specific packaging with multi-language instructions
Limited-Capability Suppliers:
- Offer only standard Hypoallergenic IV Cannula configurations
- Minimal branding options beyond basic logo printing
- Cannot accommodate special packaging requirements
- Rigid MOQ requirements regardless of customization level
Buyer Impact: Customization flexibility allows you to differentiate products, meet specific market needs, and build stronger brand recognition.
Part 2: Essential Due Diligence for Safety IV Cannula Procurement
2.1 Pre-Qualification Checklist
Verify Manufacturing Credentials:
- Request current ISO 13485 certificate with scope covering IV catheters
- Confirm FDA establishment registration and device listing
- Validate CE certificate issued by notified body (not self-declaration)
- Check for adverse event history in FDA MAUDE database
- Review customer references from established healthcare distributors
Assess Production Capacity:
- Confirm monthly production volume exceeds your projected needs
- Evaluate lead times for standard and custom Winged IV Catheter orders
- Verify backup production capabilities for supply continuity
- Review inventory management systems for just-in-time delivery
- Assess scalability for future volume growth
Evaluate Quality Systems:
- Request facility audit permission or third-party audit reports
- Review SOP documentation for Safety IV Cannula production
- Examine complaint handling procedures and CAPA processes
- Verify incoming material inspection protocols
- Check finished product release criteria and testing frequency
2.2 Sample Testing Protocol
Phase 1: Initial Samples
- Order multiple batches of Hypoallergenic IV Cannula samples
- Conduct visual inspection for manufacturing defects
- Test needle sharpness and bevel consistency
- Verify catheter flexibility and kink resistance
- Assess safety mechanism activation smoothness
Phase 2: Clinical Evaluation
- Arrange controlled clinical testing with medical professionals
- Document insertion success rates and patient feedback
- Monitor for allergic reactions or adverse events
- Compare performance against incumbent Safety IV Cannula supplier
- Evaluate packaging integrity and sterility maintenance
Phase 3: Batch Consistency Testing
- Order samples from three different production batches
- Perform comparative analysis for specification consistency
- Test across different gauge sizes in the Winged IV Catheter range
- Verify lot-to-lot color coding accuracy
- Confirm documentation consistency across batches

2.3 Pricing Structure Analysis
Beyond Unit Cost: While price is important, evaluate total cost of ownership:
- Quality Costs: Lower-priced Safety IV Cannula with higher defect rates cost more long-term
- Regulatory Costs: Suppliers without proper documentation create hidden compliance expenses
- Logistics Costs: Evaluate shipping terms, packaging efficiency, and minimum order requirements
- Service Costs: Consider technical support, regulatory assistance, and training provisions
- Risk Costs: Factor in supply chain reliability and business continuity planning
Red Flags in Pricing:
- Prices significantly below market average for Hypoallergenic IV Cannula
- Unclear pricing structure with hidden fees
- Pressure to commit to large orders without sample validation
- Unwillingness to provide written quotations with specifications
- Payment terms that require full payment before shipment
2.4 Contract Negotiation Essentials
Critical Contract Terms:
Quality Specifications:
- Detailed technical specifications for each Winged IV Catheter model
- Acceptable Quality Level (AQL) for sampling inspection
- Defect classification and acceptance criteria
- Batch rejection and replacement procedures
- Quality documentation requirements per shipment
Intellectual Property Protection:
- Confidentiality agreements for custom designs
- Trademark usage rights for branded Safety IV Cannula
- Protection against unauthorized third-party sales
- Ownership clarity for co-developed products
Supply Continuity:
- Minimum guaranteed production capacity allocation
- Lead time commitments with penalty clauses
- Alternative sourcing rights in case of supply failure
- Notification requirements for manufacturing changes
- Raw material shortage contingency plans
Liability & Insurance:
- Product liability insurance verification
- Indemnification for manufacturing defects
- Recall cooperation procedures
- Adverse event reporting responsibilities

Part 3: Building Strategic Long-Term Partnerships
3.1 Transition from Vendor to Partner
Strategic partnerships differ from transactional relationships:
Transactional Vendor Relationship:
- Price-focused negotiations
- Order-by-order engagement
- Limited communication beyond purchase orders
- Reactive problem-solving
- Supplier treats you as replaceable customer
Strategic Partnership:
- Value-based collaboration
- Joint business planning and forecasting
- Regular communication and transparency
- Proactive innovation and improvement
- Mutual commitment to long-term success
3.2 Partnership Development Framework
Phase 1: Foundation Building (Months 1-6)
Establish Communication Protocols:
- Designate dedicated account managers on both sides
- Schedule monthly business review meetings
- Create direct technical support channels
- Implement shared quality monitoring dashboards
- Develop escalation procedures for urgent issues
Align on Quality Expectations:
- Conduct joint quality standard workshops
- Agree on inspection criteria for Safety IV Cannula acceptance
- Establish collaborative CAPA processes
- Share customer feedback and market insights
- Implement continuous improvement metrics
Phase 2: Integration & Optimization (Months 6-18)
Supply Chain Integration:
- Share demand forecasts for better Hypoallergenic IV Cannula production planning
- Implement vendor-managed inventory (VMI) where appropriate
- Develop just-in-time delivery schedules
- Create safety stock strategies for critical SKUs
- Optimize packaging and shipping for cost efficiency
Co-Development Initiatives:
- Collaborate on new Winged IV Catheter design improvements
- Share market research and clinical feedback
- Joint participation in trade shows and conferences
- Co-branded marketing materials development
- Pilot testing of next-generation products
Phase 3: Strategic Expansion (18+ Months)
Market Growth Collaboration:
- Exclusive distribution agreements for new territories
- Joint investment in regulatory approvals for new markets
- Collaborative clinical studies and white papers
- Shared intellectual property development
- Volume commitment tiers with preferential pricing
Innovation Partnership:
- Early access to new Safety IV Cannula technologies
- Custom product development for niche applications
- Joint patent applications for co-created designs
- Participation in manufacturer’s innovation advisory board
- Priority allocation during supply constraints
3.3 Key Performance Indicators for Partnership Health
Track these metrics quarterly:
Quality Metrics:
- Defect rate per 100,000 units for Hypoallergenic IV Cannula
- Customer complaint frequency and resolution time
- Batch rejection rate
- Regulatory audit findings
- Product recall incidents
Delivery Performance:
- On-time delivery percentage
- Order fill rate accuracy
- Lead time consistency
- Shipping damage incidents
- Emergency order accommodation rate
Responsiveness Metrics:
- Quote turnaround time
- Technical inquiry response time
- Complaint acknowledgment speed
- Documentation provision timeliness
- Innovation proposal frequency
Financial Health:
- Price competitiveness vs. market benchmark
- Total cost of ownership trends
- Payment term flexibility
- Volume discount optimization
- Currency risk management
3.4 Relationship Governance Structure
Establish Formal Partnership Reviews:
Quarterly Business Reviews:
- Performance scorecard review across all KPIs
- Demand forecast updates and capacity planning
- Quality trend analysis and improvement initiatives
- New product development pipeline updates
- Market intelligence sharing
Annual Strategic Planning:
- Three-year business growth projections
- Investment requirements for capacity expansion
- Regulatory strategy for new market entries
- Innovation roadmap alignment
- Contract renewal negotiations
Executive Sponsorship:
- Senior leadership engagement from both organizations
- Annual executive summits to align strategic vision
- Conflict resolution escalation path
- Investment decision authority for major initiatives
3.5 Risk Mitigation Strategies
Diversification Considerations:
While building strong partnerships is valuable, smart procurement includes risk management:
80/20 Sourcing Strategy:
- Primary partner supplies 80% of Safety IV Cannula volume
- Secondary qualified supplier maintains 20% for supply security
- Regular qualification of backup suppliers
- Maintain technical files for rapid supplier switching if needed
Technology Transfer Planning:
- Document critical specifications and processes
- Maintain tooling ownership where feasible
- Periodic audits to verify manufacturing consistency
- Contingency plans for unexpected supply disruption
Contractual Protections:
- Business continuity requirements in partnership agreements
- Financial health monitoring and early warning indicators
- Exit clauses with reasonable transition periods
- Intellectual property protection for custom Winged IV Catheter designs
Part 4: Red Flags & Warning Signs
4.1 Manufacturing Concerns
Immediately disqualify suppliers who:
- Refuse factory audits or video tours
- Cannot provide verifiable certification documents
- Offer Hypoallergenic IV Cannula without biocompatibility data
- Show inconsistent product quality across samples
- Have unclear manufacturing location (trading companies posing as manufacturers)
4.2 Business Practice Concerns
Proceed with caution if supplier:
- Pressures for large upfront payments without established relationship
- Changes specifications without formal change control
- Cannot provide customer references in your market
- Shows high employee turnover in key account management roles
- Lacks transparent communication about production challenges
4.3 Regulatory Warning Signs
Critical red flags:
- Expired or suspended certifications
- Certificates that don’t match product specifications
- Unwillingness to provide regulatory technical files
- History of FDA warning letters or import alerts
- Cannot explain regulatory status in target markets

Conclusion: Your Strategic Procurement Action Plan
Selecting the right Safety IV Cannula manufacturer requires thorough due diligence, systematic evaluation, and strategic relationship building. The differences between manufacturers extend far beyond price—encompassing quality systems, innovation capabilities, regulatory compliance, and partnership potential.
Immediate Next Steps:
- Develop Your Evaluation Matrix: Create weighted scoring for quality, compliance, customization, and partnership potential
- Shortlist 3-5 Manufacturers: Apply the pre-qualification checklist to identify serious contenders
- Conduct Systematic Testing: Follow the sample testing protocol before committing to volume orders
- Negotiate Strategic Terms: Focus on long-term value, not just initial pricing
- Plan for Partnership Development: Implement the phased approach to building true strategic collaboration
Long-Term Success Factors:
The most successful Winged IV Catheter procurement programs share these characteristics:
- Clear quality specifications and expectations
- Regular performance monitoring and feedback
- Open communication and collaborative problem-solving
- Mutual investment in relationship development
- Balanced approach to cost, quality, and innovation
By applying the frameworks in this guide, you’ll move beyond transactional purchasing to build strategic partnerships that deliver superior Hypoallergenic IV Cannula products, reliable supply, and competitive advantage in your markets.



About Kohope Medical
Kohope specializes in manufacturing premium Safety IV Cannula, Winged IV Catheter, and Hypoallergenic IV Cannula solutions for global healthcare providers. With ISO 13485 certification, comprehensive customization capabilities, and a commitment to partnership-based relationships, Kohope supports distributors and healthcare organizations in delivering excellent patient care.
Ready to evaluate Kohope as your strategic Safety IV Cannula partner? Contact our team for comprehensive product information, facility documentation, and sample arrangements.