In modern medical and pharmaceutical environments, contamination control and occupational safety define operational excellence. The Blunt Filter Needle has emerged as a critical tool for mitigating particulate contamination and needlestick injury risks during medication preparation workflows.
Hospitals, compounding pharmacies, pharmaceutical manufacturers, and medical distributors increasingly specify Blunt Filter Needles for aseptic drug withdrawal from ampoules and vials. Unlike conventional sharp-tipped needles, a Blunt Needle eliminates accidental puncture hazards while maintaining optimal fluid transfer efficiency.
As an ISO-certified Needle Manufacturer, Kohope delivers precision-engineered disposable Blunt Filter Needles designed to meet stringent global medical supply chain requirements.
Understanding Blunt Filter Needle Technology
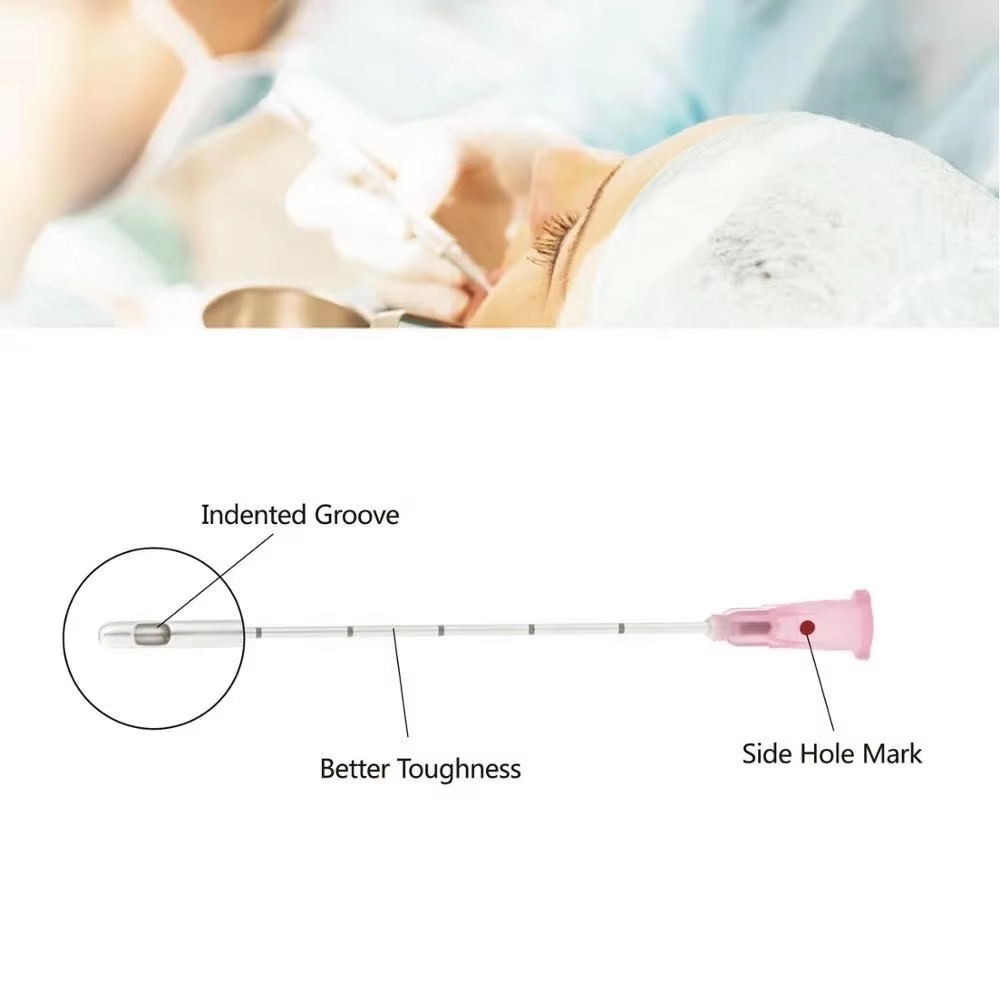
A Blunt Filter Needle represents specialized pharmaceutical equipment engineered for contamination-free medication transfer. The rounded, non-coring tip design enables safe aspiration while the integrated filtration membrane intercepts:
- Glass microparticles released during ampoule fracture
- Elastomeric fragments from rubber vial stoppers
- Airborne particulate matter introduced during preparation
This dual-function architecture addresses both physical safety and pharmaceutical quality assurance—two non-negotiable requirements in contemporary clinical practice.
Medical protocols typically mandate using a Blunt Needle during reconstitution and withdrawal stages, followed by needle exchange to a standard hypodermic for patient administration. This procedural separation optimizes both preparation safety and injection precision.

Blunt Filter Needles vs. Standard Hypodermic Needles: A Comparative Analysis
Safety Performance
Standard sharp needles present persistent occupational hazards. The CDC reports that healthcare workers experience approximately 385,000 needlestick injuries annually in the United States alone, with medication preparation accounting for significant incident frequency.
Blunt Filter Needles fundamentally eliminate penetration risk during the withdrawal phase. The rounded cannula tip cannot pierce skin or gloves, reducing injury probability by an estimated 80-95% during drug preparation activities.
Filtration Capability
Conventional needles offer zero particulate retention. Glass shards from ampoule breakage or coring fragments from repeated vial punctures enter medication solutions directly, creating contamination pathways.
Blunt Filter Needles incorporate medical-grade filtration membranes (typically 5-micron pore size) that capture submillimeter particles. This mechanical barrier ensures cleaner medication transfer without requiring additional filtration steps.
Workflow Efficiency
Traditional approaches often require separate filtration devices, increasing preparation time and consumable costs.
Integrated Blunt Needle technology consolidates safety and filtration into a single-use device. Ultra-thin wall construction maintains flow rates comparable to standard needles despite the filtration element, preventing workflow bottlenecks in high-volume environments.
Cost-Effectiveness for Institutional Buyers
While Blunt Filter Needles carry higher unit costs than basic hypodermic needles, total cost of ownership analysis reveals significant advantages:
- Reduced needlestick injury claims and associated workers’ compensation expenses
- Elimination of separate filter needle procurement
- Decreased medication waste from contamination incidents
- Simplified inventory management through consolidated SKUs
For pharmaceutical distributors evaluating supplier partnerships, these operational economics justify premium positioning in institutional procurement specifications.

Strategic Procurement Considerations for Medical Distributors
Manufacturing Quality Validation
Reputable Needle Manufacturer credentials extend beyond basic ISO certification. Procurement teams should verify:
Cleanroom production environments meeting ISO Class 7 or superior standards for sterile medical device manufacturing
Automated quality inspection systems including dimensional verification, filter membrane integrity testing, and sterility assurance protocols
Traceability infrastructure enabling lot-level tracking from raw material sourcing through final packaging
Regulatory compliance documentation covering FDA 510(k) clearances, CE marking, and region-specific medical device registrations
Kohope maintains comprehensive quality management systems with full validation documentation available for distributor audit requirements.
Specification Flexibility and Customization
Standard Blunt Filter Needle portfolios should encompass:
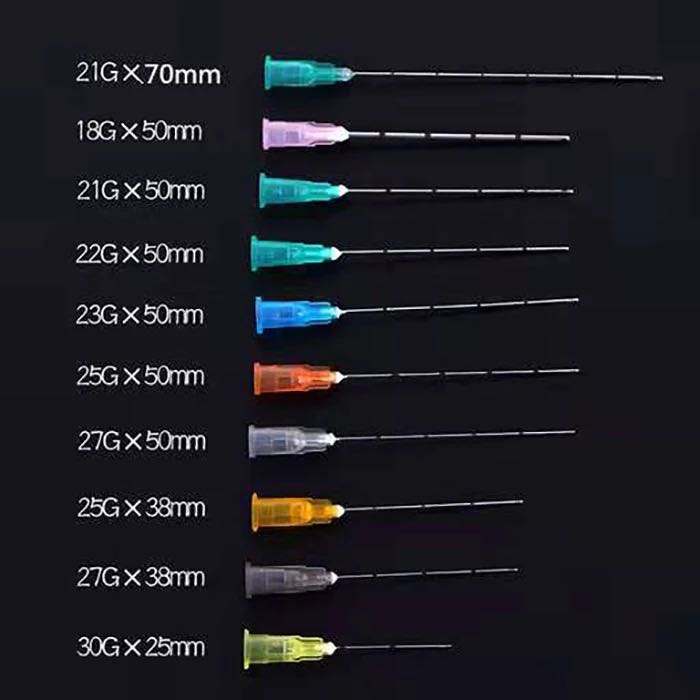
- Gauge range: 18G through 30G to accommodate viscosity variations from reconstituted antibiotics to lipid emulsions
- Length options: 25mm to 40mm configurations for different vial geometries
- Hub compatibility: Luer-lock and Luer-slip variants for syringe interoperability
- Filter specifications: Membrane materials and pore sizes suitable for specific pharmaceutical applications
Advanced Needle Manufacturer partners offer private label capabilities, custom packaging configurations, and specification modifications to support differentiated distributor positioning.
Supply Chain Reliability Metrics
Global medical supply volatility has elevated supply assurance to a primary selection criterion. Evaluate potential suppliers on:
Production capacity scalability to accommodate demand surges during public health emergencies
Raw material sourcing diversification reducing dependency on single-source stainless steel or polymer suppliers
Inventory buffer policies maintaining strategic stock for established distributor relationships
Logistics infrastructure supporting multi-region distribution with temperature-controlled shipping where required
Kohope’s manufacturing footprint and global export experience provide distributors with consistent Blunt Filter Needle availability across geopolitical disruptions.
Clinical Applications Driving Market Expansion
Hospital Pharmacy Compounding
Sterile compounding facilities increasingly mandate Blunt Filter Needles for hazardous drug preparation under USP <800> guidelines. The rounded tip design prevents core formation in chemotherapy vial stoppers, reducing particulate contamination in cytotoxic preparations.
Vaccine Preparation Workflows
Multi-dose vial protocols benefit significantly from Blunt Needle technology. Repeated punctures with sharp needles progressively damage rubber closures, releasing elastomeric particles into vaccine suspensions. Blunt-tip cannulas preserve closure integrity across sequential withdrawals.
Reconstitution of Lyophilized Medications
Antibiotics, immunoglobulins, and specialty biologics supplied as lyophilized powders require diluent addition. Blunt Filter Needles prevent glass contamination during ampoule opening while enabling efficient fluid transfer during reconstitution.
Laboratory Sample Transfer
Research and diagnostic laboratories utilize Blunt Filter Needles for contamination-sensitive sample handling, particularly when transferring viscous biologics or particulate-laden specimens between containers.
These expanding application domains position Blunt Filter Needles as essential consumables across institutional healthcare rather than niche specialty products.
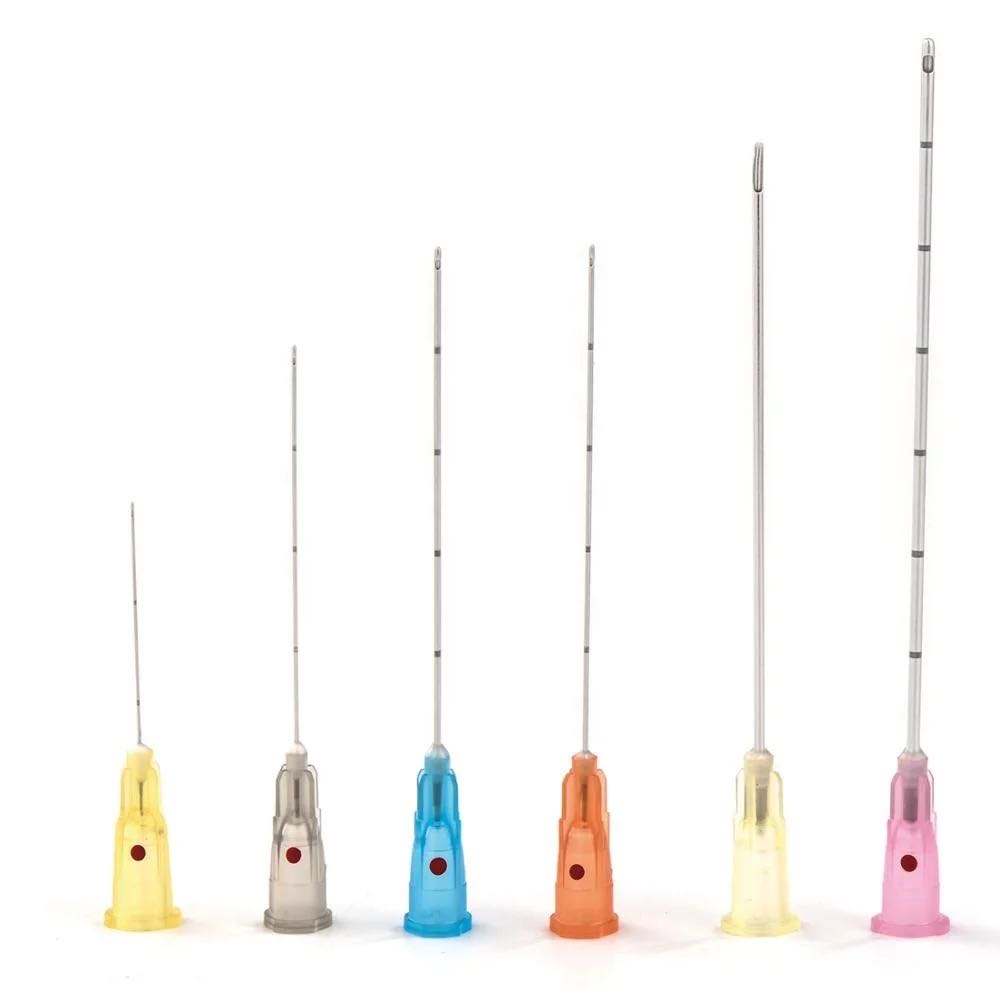
Technical Specifications That Define Performance Quality
Material Science and Biocompatibility
Premium Blunt Filter Needles utilize medical-grade 304 or 316L stainless steel cannulas, selected for:
- Corrosion resistance when exposed to acidic or alkaline pharmaceutical formulations
- Mechanical strength preventing deformation during vial stopper penetration
- Biocompatibility meeting ISO 10993 standards for tissue contact materials
Inferior alloys exhibit premature dulling, requiring excessive force during vial access and increasing preparation time.
Filter Membrane Technology
The filtration element represents the critical functional component. Professional Needle Manufacturer specifications should detail:
Membrane material composition (commonly nylon, polyethersulfone, or PTFE) selected for chemical compatibility with diverse drug formulations
Pore size distribution with tight tolerances ensuring consistent particle retention without compromising flow rate
Membrane surface area optimized to balance filtration efficiency against fluid resistance
Extractables and leachables data confirming pharmaceutical compatibility per USP <1663> guidelines
Kohope’s Blunt Filter Needles undergo rigorous pharmaceutical compatibility testing across common medication classes.
Ultra-Thin Wall Engineering
Wall thickness directly impacts flow dynamics. Standard wall needles create significant aspiration resistance, particularly with viscous medications or smaller gauge sizes.
Ultra-thin wall construction maintains external gauge diameter while maximizing internal lumen diameter. This geometry reduces aspiration force requirements by 30-40% compared to regular wall equivalents, minimizing hand fatigue during high-volume preparation sessions.
Regulatory Landscape and Compliance Requirements
Regional Medical Device Classifications
Blunt Filter Needles fall under Class II medical device regulations in most jurisdictions:
- United States: FDA 21 CFR 880.5570 (Hypodermic single lumen needle)
- European Union: MDR Annex VIII Rule 5 (Invasive devices for short-term use)
- China: NMPA Class II medical devices requiring registration certification
Distributors must verify that Needle Manufacturer partners maintain current registrations for target market regions, including ongoing post-market surveillance compliance.
Sterility Assurance and Packaging Standards
Disposable Blunt Needles require terminal sterilization validated to achieve sterility assurance levels (SAL) of 10⁻⁶ or better. Common sterilization modalities include:
- Ethylene oxide (EtO) for temperature-sensitive filter membranes
- Gamma irradiation for materials tolerating ionizing radiation exposure
Individual sterile barrier packaging must demonstrate microbial barrier properties per ISO 11607 standards, with peel-open pouches enabling aseptic presentation.

Partnership Advantages with Kohope as Your Needle Manufacturer
Comprehensive Product Portfolio
Kohope’s Blunt Filter Needle collection spans 18G through 30G specifications, accommodating applications from viscous suspension transfer to precise microvolume sampling. This gauge diversity enables distributors to serve diverse institutional requirements through a single supplier relationship.
OEM and Private Label Manufacturing
Brand differentiation increasingly drives distributor strategy in commoditized medical supply markets. Kohope supports:
- Custom packaging design incorporating distributor branding and product information
- Specification modifications tailoring needle length, hub color coding, or filter specifications to unique market requirements
- Regulatory documentation support providing technical files for distributor-led product registrations in new markets
Quality Documentation and Audit Transparency
Institutional procurement demands comprehensive validation documentation. Kohope provides:
- Certificate of Analysis (CoA) for each production lot
- Sterilization validation reports
- Biocompatibility test summaries
- Design verification and validation (V&V) documentation
- Risk management files per ISO 14971
This documentation infrastructure accelerates distributor qualification cycles and supports customer tender requirements.
Global Export Infrastructure
With established logistics partnerships and export compliance systems, Kohope facilitates seamless international distribution. Services include:
- Consolidated shipping for mixed product orders
- Documentation support for customs clearance
- Temperature-controlled transport where specifications require
- Flexible Incoterms arrangements supporting diverse distributor logistics models

Market Trends Shaping Blunt Filter Needle Demand
Infection Prevention Protocol Evolution
Healthcare-associated infection (HAI) prevention initiatives increasingly scrutinize medication preparation practices. Organizations such as the Institute for Safe Medication Practices (ISMP) now recommend Blunt Filter Needles as standard equipment for ampoule medication withdrawal, driving specification changes in hospital formularies.
Compounding Pharmacy Regulation Expansion
Following high-profile contamination incidents, regulatory oversight of compounding operations has intensified globally. USP chapters <797> (sterile compounding) and <800> (hazardous drugs) establish environmental and procedural requirements that favor Blunt Needle adoption for contamination control.
Occupational Safety Legislation
Needlestick Safety and Prevention Acts in various jurisdictions mandate healthcare facilities to evaluate and implement engineering controls that eliminate or minimize exposure. Blunt Filter Needles represent clear engineering controls during the preparation phase, creating regulatory compliance drivers beyond clinical benefit considerations.
Sustainability and Single-Use Device Optimization
While reusable medical devices gain attention for environmental reasons, medication preparation applications require single-use sterile devices. Focus has shifted toward optimized packaging (reduced plastic usage) and material selection (recyclable components where sterilization permits). Progressive Needle Manufacturer partners actively address these sustainability dimensions.

Implementation Guidance for Healthcare Facilities
Clinical Staff Training Requirements
Introducing Blunt Filter Needles requires brief procedural training focusing on:
- Proper vial stopper penetration technique with rounded tips (requiring perpendicular insertion rather than the angled approach used with sharp needles)
- Visual confirmation of filter membrane integrity before use
- Appropriate needle exchange procedures before patient administration
- Correct disposal protocols in sharps containers despite reduced puncture risk
Most facilities achieve full staff competency within a single training session, minimizing implementation friction.
Inventory Management Optimization
Transitioning to Blunt Filter Needles allows inventory rationalization by consolidating what previously required separate sharp needles and filter devices. Pharmacy departments typically establish:
- Par levels based on average daily medication preparation volume
- Gauge distribution weighted toward commonly used sizes (typically 18G and 21G account for 60-70% of consumption)
- Automated reorder triggers through materials management systems
Cost-Benefit Analysis Framework
Institutional adoption decisions benefit from structured financial evaluation incorporating:
Direct costs: Blunt Filter Needle unit price versus combined cost of standard needles plus separate filter needles
Avoided costs: Needlestick injury medical evaluation, testing, prophylaxis, and workers’ compensation claims (average cost per incident: $3,000-$7,000)
Quality costs: Medication waste from contamination detection and associated preparation rework
Productivity gains: Time savings from integrated filtration eliminating additional process steps
Comprehensive models consistently demonstrate positive return on investment within 12-18 months for medium to large hospital pharmacy operations.

Conclusion: Strategic Value of Blunt Filter Needle Adoption
The convergence of patient safety imperatives, occupational health requirements, and pharmaceutical quality standards has positioned Blunt Filter Needles as fundamental infrastructure in modern medication preparation workflows.
For medical distributors and pharmaceutical suppliers, these market dynamics create compelling portfolio expansion opportunities. Success requires partnership with a Needle Manufacturer demonstrating:
- Consistent manufacturing quality validated through comprehensive documentation
- Product specification breadth supporting diverse institutional requirements
- Supply chain reliability withstanding global disruption scenarios
- Customization capabilities enabling market differentiation
- Technical support infrastructure accelerating customer implementation
Kohope’s two decades of precision medical device manufacturing experience, combined with dedicated export capabilities and quality management systems, position us as a strategic partner for distributors seeking to capitalize on expanding Blunt Needle market opportunities.
Whether you require bulk wholesale supply, OEM manufacturing, or custom specification development, our team provides responsive technical consultation and streamlined procurement processes designed specifically for B2B partnerships.
Explore our complete Blunt Filter Needle product specifications or contact our distribution team to discuss volume pricing and partnership opportunities.