Diabetes management has evolved significantly over the past decades, yet one tool remains fundamental to millions of patients worldwide: the insulin syringe. As medical device manufacturing advances, understanding the precision engineering and quality control behind these essential instruments becomes crucial for healthcare providers, procurement specialists, and patients alike.
Understanding Insulin Syringe Manufacturing Technology
The production of insulin syringes represents a sophisticated intersection of polymer science, precision molding, and medical device manufacturing protocols. Modern insulin syringes require exceptional dimensional accuracy to ensure proper dosage delivery—a matter where even microliter variations can impact patient outcomes.
Core Manufacturing Processes
Injection Molding Precision
The barrel and plunger components begin as medical-grade polypropylene or polyethylene resins. High-precision injection molding machines maintain tolerances within ±0.01mm to ensure consistent barrel diameter and smooth plunger movement. The molding process occurs in cleanroom environments classified to ISO Class 7 or 8 standards, minimizing particulate contamination from the outset.
Temperature control during molding is critical—resin temperatures typically range from 200-240°C depending on polymer composition, while mold temperatures are maintained between 40-80°C. This thermal management prevents warping and ensures dimensional stability across millions of units.
Graduated Scale Application
The distinctive black graduation markings on insulin syringe barrels use pad printing or screen printing technologies with medical-grade inks. For U-100 insulin (the international standard concentration of 100 units per milliliter), markings must achieve precision within 2% of nominal volume. Advanced vision inspection systems verify every graduation during production, automatically rejecting units outside specification.
Needle Integration Technology
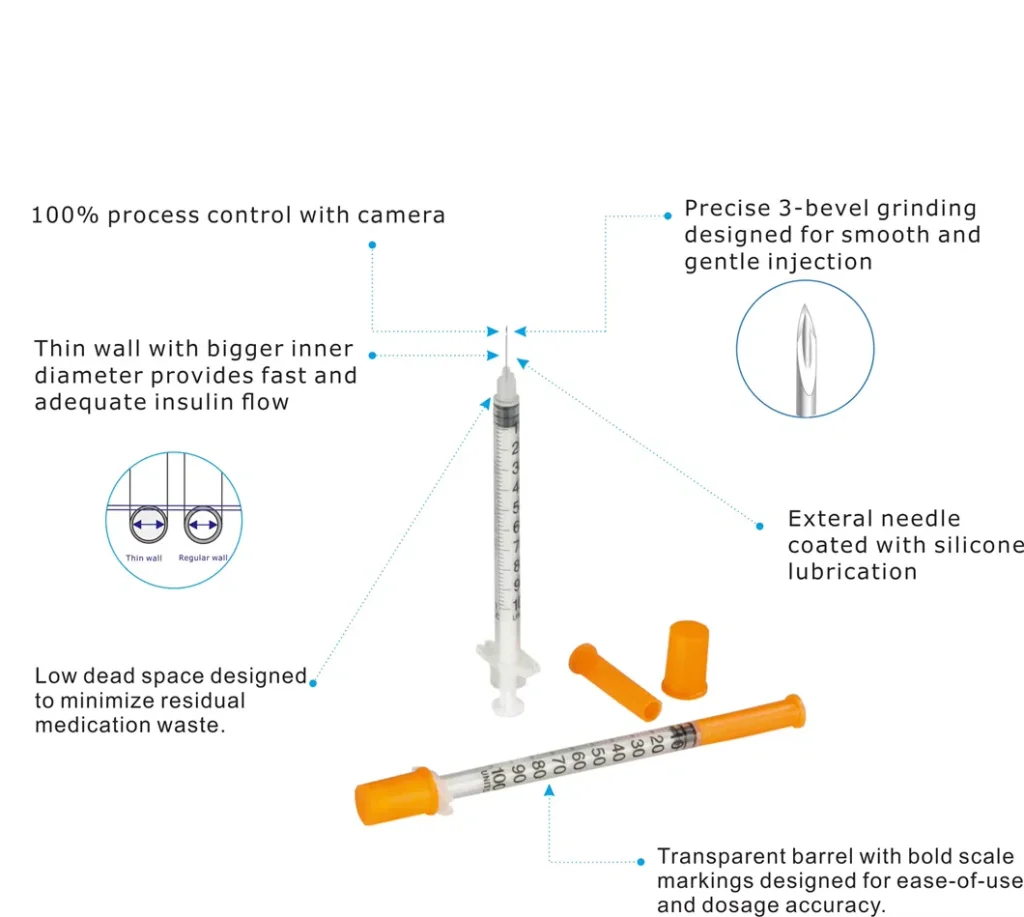
Needles undergo separate manufacturing before integration. Stainless steel tubing (typically 304 or 316L grade) is precision-cut, ground to create the bevel angle, and electropolished to achieve smooth surfaces that minimize tissue trauma. Needle gauges from 28G to 32G correspond to outer diameters ranging from 0.36mm down to 0.23mm—dimensions requiring specialized grinding equipment.
For fixed-needle designs, ultrasonic welding or adhesive bonding permanently attaches the needle hub to the barrel. This connection must withstand pull forces exceeding 3N while maintaining a hermetic seal. Detachable needle designs employ precision-molded luer locks with standardized ISO 594 threading.

Dead Space Minimization Engineering
One of the most critical technical achievements in modern premium disposable insulin syringes is minimizing dead space—the residual volume remaining after injection. Traditional syringes might retain 0.03-0.05ml, but advanced designs reduce this to under 0.01ml through precision engineering of the plunger tip geometry and barrel interior finish.
This reduction translates directly to cost savings for patients and reduced insulin waste, particularly important for expensive analog insulins. The plunger tip typically features a conical or dome shape, manufactured with surface roughness values below Ra 0.8μm to ensure complete fluid displacement.
Quality Control and Regulatory Compliance
Manufacturing insulin syringes under ISO 13485 certification requires exhaustive quality management systems. Production facilities implement multi-tier inspection protocols:
In-Process Quality Checkpoints
- Dimensional verification using coordinate measuring machines (CMM) with resolution to 0.001mm
- Automated optical inspection detecting defects including flash, short shots, or contamination
- Plunger force testing ensuring smooth, consistent operation within 5-15N range
- Needle sharpness testing through penetration force measurement
- Dead volume quantification via precision fluid displacement
Sterilization Validation
Ethylene oxide (EO) sterilization remains the standard method for insulin syringes due to material compatibility. The process typically involves:
- Pre-conditioning at 30-40°C and 60-80% relative humidity
- EO exposure (concentration 450-1200 mg/L) for 2-6 hours at 40-55°C
- Aeration period extending 8-16 hours to reduce residual EO below 10 ppm
Biological indicators using Bacillus atrophaeus spores verify sterility assurance level (SAL) of 10⁻⁶, meaning less than one in one million probability of non-sterility.
Biocompatibility Testing
Materials undergo rigorous testing per ISO 10993 standards, including cytotoxicity, sensitization, and irritation studies. Latex-free and BPA-free formulations address allergic concerns, expanding patient safety profiles.
Clinical Applications and Usage Scenarios
Daily Diabetes Management
Approximately 537 million adults worldwide live with diabetes, with many requiring daily insulin therapy. Insulin syringes serve as the primary delivery method for:
Type 1 Diabetes Patients: These individuals require multiple daily injections (MDI) of rapid-acting insulin before meals and long-acting insulin once or twice daily. The 0.3ml and 0.5ml syringe sizes suit pediatric and lean adult patients with smaller dose requirements, while 1ml syringes accommodate larger doses for insulin-resistant patients.
Type 2 Diabetes with Insulin Requirement: When oral medications prove insufficient, insulin therapy becomes necessary. Many Type 2 patients begin with basal insulin once daily, making the precise dosing of insulin syringes essential for achieving target glycemic control without hypoglycemia risk.
Gestational Diabetes Management: Pregnant women developing diabetes often require insulin therapy to protect fetal development while maintaining maternal health. The safety profile and dosing accuracy of disposable insulin syringes make them ideal for this temporary but critical treatment period.
Hospital and Clinical Settings
Endocrinology Departments: Specialized diabetes centers utilize insulin syringes for inpatient diabetes management, particularly during illness, surgery, or metabolic crisis when blood glucose becomes unstable.
Emergency Medicine: Diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS) require rapid insulin administration. While intravenous insulin infusions often initiate treatment, subcutaneous insulin injections via syringes support transition to maintenance therapy.
Perioperative Care: Surgical patients with diabetes require careful glucose management. Insulin syringes enable flexible dosing adjustments based on frequent blood glucose monitoring during the perioperative period.
Pharmacy and Retail Distribution
Community pharmacies serve as primary access points for diabetes supplies. Premium disposable insulin syringes must meet both clinical efficacy and patient comfort requirements:
- 28G needles (0.36mm diameter): Standard choice balancing injection comfort with flow rate
- 30G needles (0.30mm diameter): Reduced tissue trauma for pain-sensitive patients
- 31-32G needles (0.25-0.23mm diameter): Ultra-fine options for pediatric use or patients with needle phobia
Needle length selection also impacts patient experience: 8mm needles suit most adults, while 6mm or shorter needles benefit children, lean individuals, or those injecting into areas with less subcutaneous tissue.
Public Health and NGO Programs
Global health initiatives addressing diabetes in underserved populations rely on affordable, reliable insulin delivery systems. Manufacturing scalability and quality consistency enable:
- Government procurement programs supplying national healthcare systems
- International relief organizations providing diabetes care in resource-limited settings
- Corporate social responsibility programs expanding diabetes access in developing regions
These applications demand not only cost efficiency but also packaging durability for distribution across challenging climates and supply chains.

Advanced Features in Modern Insulin Syringe Design
Ergonomic Plunger Engineering
Contemporary designs incorporate finger grips with textured surfaces, improving control during injection and reducing the risk of dosing errors. The plunger stopper material—typically bromobutyl rubber or synthetic elastomers—must balance flexibility for smooth movement with rigidity to prevent deformation under pressure.
Barrel Transparency and Marking Visibility
Crystal-clear barrel materials with high light transmission (>90%) allow patients to detect air bubbles and verify insulin appearance. Bold black graduations provide contrast for patients with visual impairments, a critical consideration given diabetic retinopathy affects many long-term diabetes patients.
Safety-Engineered Options
While traditional insulin syringes lack needle safety features due to cost considerations, newer designs incorporate retractable needles or protective shields for healthcare workers at risk of needlestick injuries. These safety-engineered devices meet OSHA bloodborne pathogen standards while maintaining the precision dosing essential for insulin therapy.
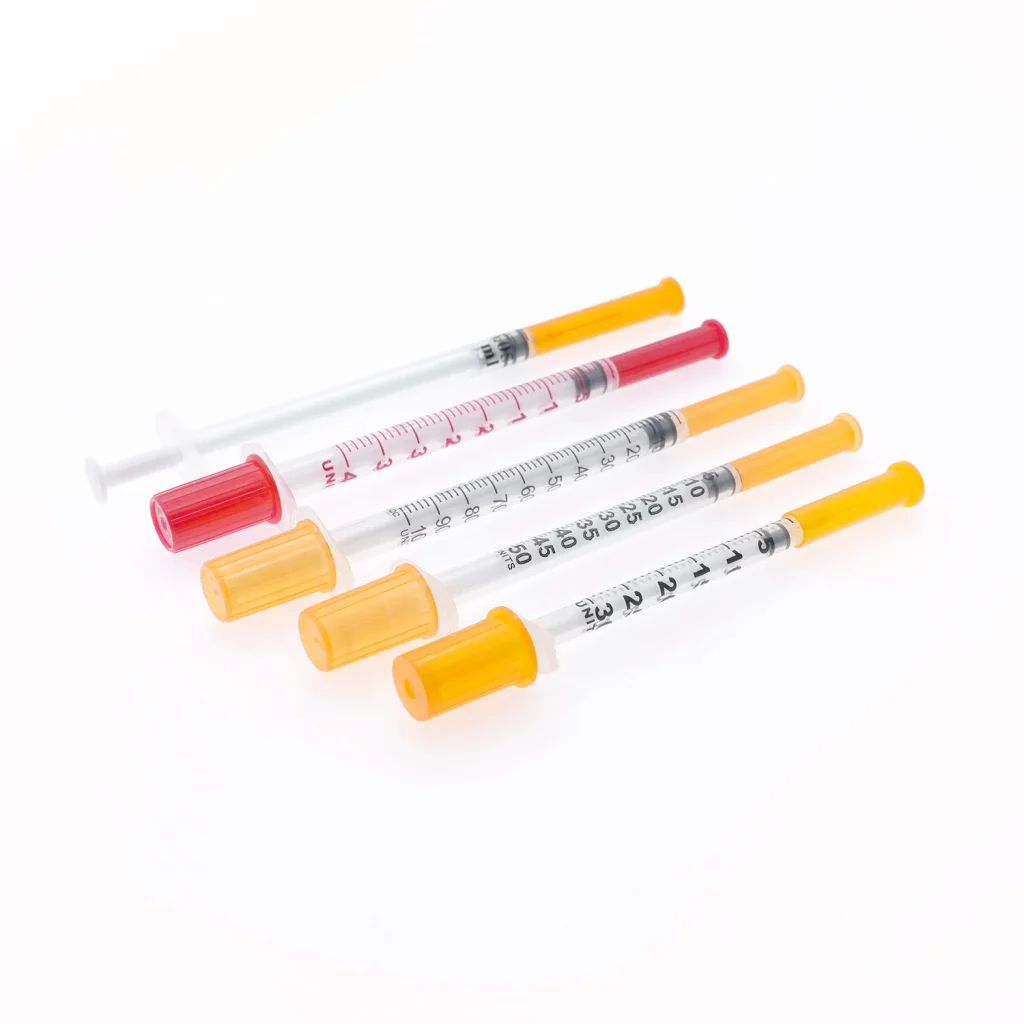
OEM/ODM Customization Capabilities
Leading manufacturers offer extensive customization for private label brands, healthcare systems, and pharmaceutical companies:
Barrel Printing: Corporate logos, brand names, or instructional graphics can be printed directly on syringe barrels using medical-grade inks resistant to alcohol swabs and handling.
Packaging Design: Blister packs, pouches, or bulk packaging adapt to different distribution models. Multilingual instructions support international markets where product information must comply with local regulatory requirements.
Specification Modification: Needle gauge combinations, barrel capacities, and graduation scales can be tailored to specific clinical protocols or patient populations.
Quality Assurance and Testing Protocols
Manufacturers committed to excellence implement comprehensive testing beyond regulatory minimums:
Mechanical Performance Testing
- Breakage force testing ensuring needle hub withstands >3N pull
- Plunger sliding force maintaining consistency across temperature ranges
- Leakage testing at pressures exceeding clinical use conditions
Functional Verification
- Dosing accuracy confirmation across full volume range
- Needle penetration force measurement simulating injection into skin
- Barrel transparency assessment ensuring bubble visibility
Stability Studies
- Accelerated aging protocols predicting 3-5 year shelf life
- Transportation simulation testing package integrity under shipping stresses
- Sterilization efficacy validation across production batches

The Future of Insulin Syringe Technology
Innovation continues advancing insulin delivery precision and patient comfort:
Material Science Developments: Research into lower-friction barrel coatings reduces injection force, benefiting patients with limited hand strength or arthritis.
Smart Integration: While maintaining affordability, some manufacturers explore incorporating dose-logging technology or connectivity features that help patients track injection adherence.
Sustainability Initiatives: Biodegradable materials and recycling programs address environmental concerns while maintaining sterility and performance standards.

Selecting the Right Manufacturing Partner
Healthcare organizations, pharmacy chains, and medical device distributors should evaluate potential suppliers based on:
- Regulatory Certifications: ISO 13485, CE marking, FDA registration (if applicable)
- Production Capacity: Ability to meet volume requirements with consistent lead times
- Quality Systems: Documented procedures for defect prevention and corrective action
- Technical Support: Engineering expertise for specification customization
- Supply Chain Reliability: Raw material sourcing stability and inventory management
Conclusion
The humble insulin syringe represents sophisticated medical device engineering serving a critical global health need. From precision molding and sterile manufacturing to clinical application across diverse healthcare settings, these devices embody the intersection of manufacturing excellence and patient care.
As diabetes prevalence continues rising worldwide, demand for high-quality, affordable insulin syringes will only increase. Manufacturers maintaining rigorous quality standards, embracing technological innovation, and supporting global access initiatives will remain essential partners in diabetes care delivery.
For healthcare providers, procurement specialists, and diabetes care programs seeking reliable insulin delivery solutions, partnering with experienced manufacturers offering ISO-certified production, customization capabilities, and consistent quality represents an investment in patient outcomes and program success.
For technical specifications, sample requests, or partnership inquiries regarding premium disposable insulin syringes, contact qualified manufacturers who can provide documentation of regulatory compliance, quality certifications, and clinical performance data.