Reading Time: 12 minutes
Introduction: The Critical Role of Auto-Disable Syringes in Modern Healthcare
In the global fight against healthcare-associated infections and needlestick injuries, auto-disable syringes have emerged as one of the most significant innovations in medical device technology. These specialized single-use injection devices incorporate an integrated self-destruct mechanism that permanently disables the syringe after a single use, effectively eliminating the possibility of reuse and dramatically reducing cross-contamination risks.
According to the World Health Organization (WHO), unsafe injection practices account for approximately 1.3 million early deaths annually, with an estimated 16 billion injections administered worldwide each year. Alarmingly, studies indicate that up to 40% of injections in developing countries are administered with syringes or needles reused without sterilization. This staggering statistic underscores the urgent need for premium auto-disable syringes that physically prevent reuse.
In this comprehensive guide, we’ll explore the technical mechanisms behind auto-disable technology, examine market trends driving adoption, analyze key application scenarios, and provide insights for healthcare organizations and distributors seeking to implement these life-saving devices.
Understanding Auto-Disable Syringe Technology: How It Works
The Fundamental Mechanism
Auto-disable syringes operate on a deceptively simple yet ingeniously effective principle: once the plunger is fully depressed to deliver the medication, an internal mechanism permanently locks or breaks, rendering the device unusable for subsequent injections.
There are two primary technical designs:
1. Front Lock (Plunger Lock) Mechanism
The front lock system features a specially engineered plunger that, upon complete depression, engages with a locking component inside the barrel. When the healthcare worker attempts to withdraw the plunger after injection, the mechanism locks into place, preventing any further retraction. This design creates an irreversible connection between the plunger and barrel, making it physically impossible to draw up another dose.
The front lock design is particularly advantageous for:
- Vaccination campaigns requiring visible proof of single-use
- Training scenarios where visual confirmation of the lock is beneficial
- Applications where immediate tactile feedback is important
2. Back Lock (Barrel Break) Mechanism
The back lock system incorporates a pre-weakened section in the plunger shaft. As the plunger reaches full depression, this weakened area fractures, causing the plunger rod to physically break apart. This catastrophic failure of the plunger structure makes reuse absolutely impossible, as the broken components cannot generate the vacuum pressure needed to draw up liquid.
Back lock mechanisms excel in:
- High-volume immunization programs where speed is critical
- Settings with less trained personnel, as the mechanism is foolproof
- Environments where syringe disposal verification is important
Both systems comply with ISO 7886-3 standards, which specifically govern the performance requirements for single-use syringes with protective features to prevent reuse.
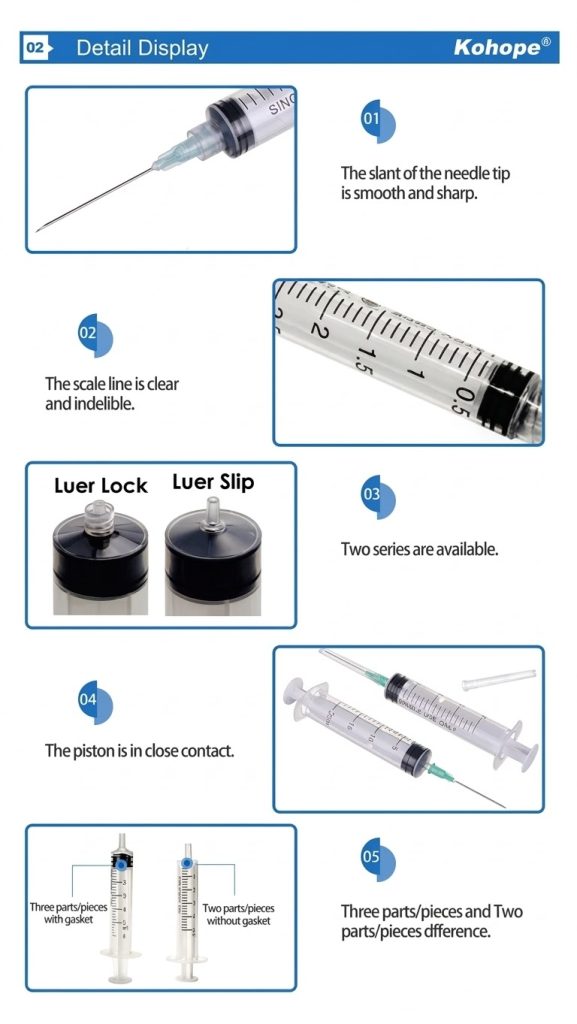
Material Science and Engineering Precision
The effectiveness of auto-disable medical syringes depends heavily on precision engineering and material selection. Medical-grade polypropylene (PP) is the material of choice for both the barrel and plunger due to its:
- Optical clarity: Allows healthcare workers to monitor medication flow and detect air bubbles
- Chemical resistance: Maintains integrity when exposed to various medications
- Dimensional stability: Ensures consistent performance across temperature variations
- Biocompatibility: ISO 10993 certified for safe patient contact
The piston component deserves special attention. Traditional natural rubber pistons provide excellent sealing properties but pose allergy risks for latex-sensitive patients. Modern auto-disable syringe manufacturers now offer synthetic rubber (isoprene rubber – IR) alternatives that deliver comparable performance without latex proteins, expanding the device’s safe application across all patient populations.
The hypodermic needle, manufactured from premium-grade stainless steel according to ISO 7864:1993 standards, features:
- Tri-bevel cutting edge: Minimizes tissue trauma and patient discomfort
- Optimized inner diameter: Ensures appropriate flow rates for different medications
- Color-coded hubs: Facilitates quick gauge identification (21G-27G range)
- Silicone coating: Reduces insertion force and enhances patient comfort
Quality Assurance and Sterilization
Manufacturing high-quality auto-disable syringes requires rigorous quality control at every production stage. Ethylene oxide (EO) gas sterilization remains the gold standard for these devices, as it:
- Penetrates all packaging layers and device components
- Operates at lower temperatures, preserving material integrity
- Effectively eliminates bacteria, viruses, and spores
- Leaves no toxic residues when proper aeration protocols are followed
Post-sterilization testing includes:
- Sterility assurance level (SAL) verification to 10⁻⁶
- Pyrogenicity testing using Limulus Amebocyte Lysate (LAL) assay
- Functional performance testing of the auto-disable mechanism
- Package integrity assessment
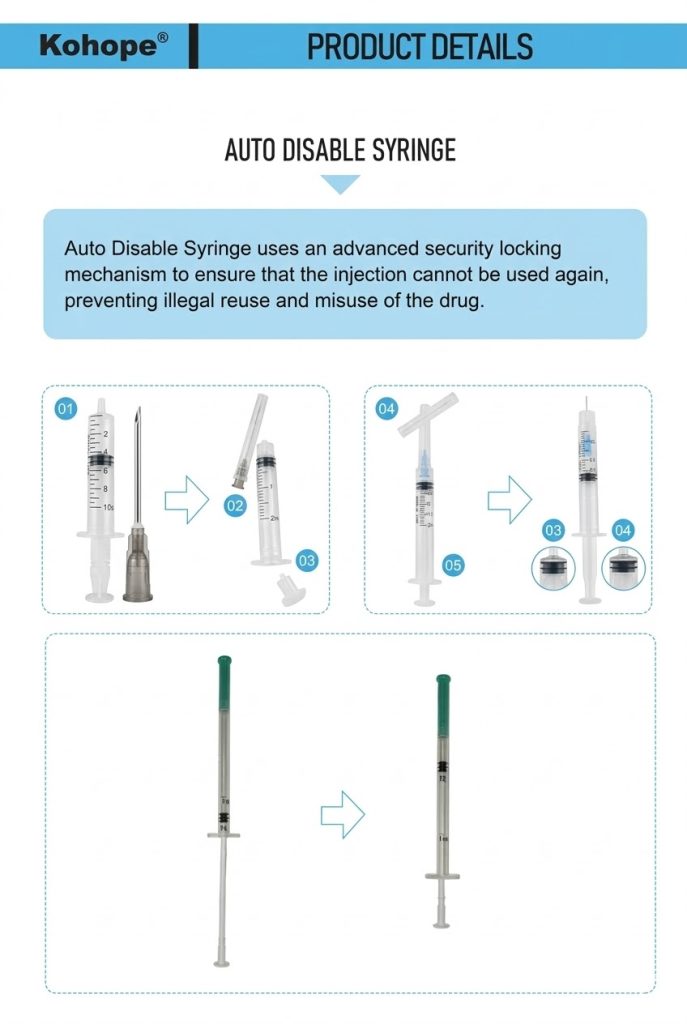
Market Dynamics: The Growing Demand for Auto-Disable Syringes
Global Market Size and Growth Projections
The global auto-disable syringes market has experienced remarkable expansion over the past decade. Valued at approximately $4.8 billion in 2023, market analysts project the sector will reach $8.2 billion by 2030, representing a compound annual growth rate (CAGR) of 7.9%.
Several factors drive this growth trajectory:
1. Regulatory Mandates and Policy Changes
Governments and international health organizations increasingly mandate auto-disable syringe use for specific applications:
- The WHO recommends exclusive use of auto-disable syringes for immunization by 2020 (achieved in most developed nations)
- India’s Mission Indradhanush program requires AD syringes for all government vaccination initiatives
- UNICEF procurement specifications prioritize auto-disable technology
- The Global Alliance for Vaccines and Immunization (GAVI) supports AD syringe adoption in 73 countries
2. Mass Vaccination Campaigns
The COVID-19 pandemic dramatically accelerated auto-disable syringe adoption. During 2021-2022, global vaccine administration exceeded 13 billion doses, with the majority utilizing auto-disable technology to ensure safety and prevent diversion of vaccines to black markets.
Beyond COVID-19, ongoing vaccination programs target:
- Polio eradication (particularly in Afghanistan and Pakistan)
- Measles-rubella elimination campaigns
- Rotavirus and pneumococcal disease prevention
- Seasonal influenza programs in aging populations
3. Healthcare Worker Safety Initiatives
Needlestick injuries affect approximately 3 million healthcare workers annually worldwide, with the highest incidence in low- and middle-income countries. Each needlestick injury carries transmission risks for:
- Hepatitis B (30% transmission probability)
- Hepatitis C (1.8% transmission probability)
- HIV (0.3% transmission probability)
Auto-disable syringes significantly reduce these risks by eliminating reuse and reducing handling of used devices.

Regional Market Variations
Asia-Pacific Region
The Asia-Pacific market represents the largest and fastest-growing segment, driven by:
- Population density requiring extensive vaccination infrastructure
- Government investments in universal immunization programs
- Manufacturing capacity expansion in India, China, and Southeast Asia
- Price-sensitive markets favoring cost-effective AD solutions
Africa and Middle East
These regions show strong growth potential due to:
- International aid programs prioritizing injection safety
- High prevalence of bloodborne diseases necessitating prevention
- Infrastructure development in healthcare delivery systems
- WHO and UNICEF procurement programs
Europe and North America
Mature markets characterized by:
- Stringent regulatory requirements and compliance standards
- Premium pricing for advanced safety features
- Focus on sustainable and eco-friendly medical devices
- Integration with electronic health record systems
Latin America
Emerging market dynamics include:
- Government initiatives to modernize healthcare infrastructure
- Growing middle class demanding quality healthcare
- Regional manufacturing development to reduce import dependence

Key Application Scenarios: Where Auto-Disable Syringes Make the Difference
1. National Immunization Programs
Auto-disable syringes have become the backbone of national immunization programs worldwide. Their application in this sector offers multiple advantages:
Vaccine Integrity Protection: The single-use design prevents vaccine dilution or contamination that occurs when syringes are reused or “topped up” with additional doses from multi-dose vials.
Supply Chain Transparency: Governments can accurately track vaccine administration by correlating vaccine doses with AD syringe distribution, reducing wastage and preventing black market diversion.
Community Confidence: Visible auto-disable mechanisms reassure parents and patients that sterile, safe injection practices are being followed, improving vaccination compliance rates.
Case Study: India’s implementation of AD syringes across its Universal Immunization Program resulted in:
- 23% reduction in injection-associated adverse events
- 94% parent satisfaction with visible safety features
- 17% improvement in vaccination coverage in rural areas
2. Insulin Administration and Diabetes Management
While traditional insulin syringes remain common, specialized auto-disable insulin syringes address specific challenges:
Preventing Insulin Pen Needle Sharing: In institutional settings (nursing homes, assisted living facilities), AD technology prevents cross-contamination from shared insulin delivery devices.
Pediatric Diabetes Programs: School-based diabetes management benefits from AD syringes that prevent accidental reuse and provide clear disposal protocols.
Emergency Glucose Administration: Pre-filled AD syringes containing glucagon or dextrose ensure single-use in emergency response scenarios.
3. Emergency Medical Services and Disaster Response
First responders and disaster relief organizations increasingly rely on auto-disable medical syringes for:
Field Triage: AD syringes with pre-attached needles enable rapid medication administration without concerns about accidental reuse in chaotic environments.
Resource-Constrained Settings: During natural disasters or humanitarian crises where sterilization infrastructure is compromised, AD syringes provide guaranteed single-use safety.
Multi-Casualty Incidents: Color-coded AD syringes facilitate triage protocols (different sizes/colors for different medications or patient categories).
4. Controlled Substance Administration
Hospitals and addiction treatment facilities use auto-disable syringes for controlled medications:
Preventing Drug Diversion: The self-destruct mechanism prevents extraction of residual medication for illicit use.
Methadone and Buprenorphine Programs: Addiction treatment centers use AD syringes to ensure prescribed doses cannot be diverted or reused.
Hospital Formulary Management: AD syringes for high-value medications (chemotherapy, biologics) prevent tampering and ensure proper dosing.
5. Veterinary Medicine
Animal healthcare applications benefit from AD technology:
Livestock Vaccination: Large-scale agricultural operations use AD syringes to prevent disease transmission within herds during mass vaccination campaigns.
Companion Animal Care: Veterinary clinics prevent cross-contamination between patients, particularly important for immunocompromised animals.
Wildlife Conservation: Field biologists use AD syringes for vaccine delivery and sampling in endangered species programs, ensuring no pathogen transmission.
6. Clinical Research and Trials
Pharmaceutical research utilizes auto-disable syringes for:
Dose Accountability: AD syringes provide verifiable proof that each trial participant received exactly one dose, critical for regulatory compliance.
Blinding Protocols: Pre-filled, identical AD syringes maintain double-blind trial integrity.
Bioavailability Studies: Single-use design eliminates variables associated with syringe cleaning or reuse that could affect pharmacokinetic measurements.
Technical Specifications: Choosing the Right Auto-Disable Syringe
Capacity and Volume Options
Modern auto-disable syringe manufacturers offer comprehensive capacity ranges:
- 0.5mL – 1mL: Ideal for vaccines, insulin, and pediatric medications requiring precise small doses
- 2mL – 3mL: Standard size for intramuscular injections, antibiotics, and routine medications
- 5mL – 10mL: Suitable for larger volume medications, contrast agents, and emergency drugs
- 20mL – 50mL: Specialized applications including irrigation, feeding tube medication delivery
The 10mL auto-disable syringe represents the optimal balance for most clinical applications, offering sufficient capacity for standard medications while maintaining ease of handling.

Needle Gauge Selection
Proper needle gauge selection impacts both patient comfort and medication delivery:
| Gauge | Inner Diameter | Typical Applications |
|---|---|---|
| 21G | 0.82mm | Blood donation, rapid fluid administration |
| 22G | 0.72mm | Intramuscular vaccines, blood draws |
| 23G | 0.64mm | Standard intramuscular injections |
| 25G | 0.51mm | Subcutaneous injections, pediatric use |
| 26G | 0.46mm | Insulin delivery, intradermal testing |
| 27G | 0.41mm | Pediatric vaccines, cosmetic procedures |
Graduation Markings and Accuracy
Medical-grade auto-disable syringes feature indelible ink graduation markings with:
- Accuracy tolerance: ±5% for volumes ≥2mL, ±10% for volumes <2mL
- Bold scale visibility: Easy reading under various lighting conditions
- Dual-scale options: mL and units for insulin administration
- Clear numbering: Every major graduation labeled for error reduction
Regulatory Landscape and Compliance Requirements
International Standards
Auto-disable syringe manufacturers must comply with multiple international standards:
ISO 7886-3:2020: Sterile hypodermic syringes for single use – Part 3: Auto-disable syringes for fixed-dose immunization
- Specifies performance requirements
- Defines testing protocols for auto-disable mechanism functionality
- Establishes labeling and marking requirements
ISO 7886-1:2017: General requirements for hypodermic syringes
- Material biocompatibility standards
- Dimensional tolerances
- Sterility assurance requirements
ISO 7864:1993: Sterile hypodermic needles for single use
- Needle sharpness specifications
- Hub attachment strength requirements
- Sterility and packaging standards
Regional Regulatory Approvals
FDA (United States)
- 510(k) premarket notification for Class II medical devices
- Good Manufacturing Practice (GMP) compliance
- Quality System Regulation (QSR) adherence
CE Marking (European Union)
- Medical Device Regulation (MDR) 2017/745 compliance
- Notified Body assessment and certification
- Post-market surveillance requirements
NMPA (China)
- Product registration for Class II medical devices
- Manufacturing site inspection
- Clinical evaluation documentation
WHO Prequalification
- Essential for UNICEF and GAVI procurement
- Demonstrates suitability for resource-limited settings
- Requires ongoing surveillance and quality monitoring
Economic Considerations: Cost-Benefit Analysis
Price Comparison
Auto-disable syringes typically cost 10-40% more than standard disposable syringes:
- Standard 3mL disposable syringe: $0.03-$0.08 USD
- 3mL auto-disable syringe: $0.05-$0.12 USD
- Premium AD syringe with safety features: $0.10-$0.18 USD
However, this upfront cost must be evaluated against:
Hidden Costs of Standard Syringes
Reuse-Related Infections: The WHO estimates that preventing unsafe injections saves $535 per hepatitis B infection avoided and $5,426 per HIV infection avoided.
Needlestick Injury Management: Each needlestick injury costs healthcare systems:
- Immediate medical evaluation: $500-$1,000
- Prophylactic treatment (if needed): $1,000-$3,000
- Follow-up testing over 6-12 months: $500-$800
- Lost productivity and anxiety: $2,000-$5,000
- Total average cost: $4,000-$10,000 per incident
Litigation and Liability: Healthcare facilities face potential lawsuits from transmission events, with settlement costs often exceeding $100,000.
Return on Investment
Large healthcare organizations implementing auto-disable medical syringes report:
- 60-85% reduction in needlestick injuries
- 95-100% elimination of syringe reuse incidents
- 30-50% reduction in injection-associated infection rates
- ROI achievement within 18-36 months for most facilities
Environmental Considerations and Sustainability
Waste Management Challenges
The global shift toward auto-disable syringes has environmental implications:
Volume: With 16 billion injections annually, medical sharps waste represents a significant disposal challenge.
Current Solutions:
- Incineration (most common, but produces air pollutants)
- Autoclaving followed by landfill (reduces infectious risk)
- Chemical disinfection (suitable for small-scale operations)
- Encapsulation in concrete or high-density materials
Sustainable Manufacturing Initiatives
Progressive auto-disable syringe manufacturers are implementing:
Reduced Plastic Formulations: Optimizing barrel thickness to minimize material use while maintaining structural integrity (10-15% material reduction possible).
Recycled Content: Investigating medical-grade recycled polypropylene for non-patient-contact components.
Biodegradable Alternatives: Research into bio-based polymers that maintain performance characteristics while offering improved end-of-life degradation.
Carbon-Neutral Manufacturing: Solar power integration, waste heat recovery, and supply chain optimization to reduce production carbon footprint.
Future Trends and Innovations
Smart Auto-Disable Syringes
Next-generation devices integrate digital technology:
RFID Tracking: Embedded chips enable:
- Real-time inventory management
- Automated cold chain monitoring for vaccines
- Counterfeit prevention
- Dose administration verification for electronic health records
Temperature Indicators: Color-changing labels alert users if vaccines have been exposed to temperature excursions.
Integrated Sharps Disposal: Syringes with built-in needle shields that automatically activate after injection, reducing needlestick risks during disposal.
Pre-Filled Auto-Disable Systems
Pharmaceutical companies increasingly offer pre-filled auto-disable syringes that combine:
- Factory-filled medication (ensures dosing accuracy)
- Integrated auto-disable mechanism
- Reduced preparation time and medication errors
- Extended shelf life with specialized barrier plastics
Customization and OEM Opportunities
Auto-disable syringe manufacturers now offer extensive customization:
- Custom barrel colors for brand differentiation
- Specialized scale markings for specific medications
- Modified plunger designs for single-handed operation
- Integrated safety features (retractable needles + auto-disable)
Conclusion: The Imperative for Auto-Disable Adoption
As global healthcare systems confront the dual challenges of improving patient safety while managing costs, auto-disable syringes represent not merely an incremental improvement but a fundamental paradigm shift in injection safety.
The evidence is compelling: these devices dramatically reduce disease transmission, protect healthcare workers, prevent medication diversion, and ultimately save lives. While the upfront cost exceeds traditional disposable syringes, the comprehensive cost-benefit analysis—factoring in infection prevention, litigation avoidance, and workforce protection—demonstrates clear economic advantages.
For healthcare administrators, procurement officers, and public health officials evaluating premium auto-disable syringe solutions, the decision framework should consider:
- Regulatory compliance: Ensure chosen products meet all applicable ISO, FDA, CE, and WHO standards
- Clinical application fit: Match syringe specifications (capacity, needle gauge, mechanism type) to specific use cases
- Supplier reliability: Partner with established manufacturers offering consistent quality, regulatory documentation, and responsive support
- Total cost of ownership: Evaluate comprehensive costs including infection prevention and safety improvements
- Sustainability: Consider environmental impact and manufacturer commitment to responsible practices
The transition to universal auto-disable syringe adoption represents an achievable goal with profound public health implications. As manufacturing capacity expands, costs decline, and regulatory frameworks strengthen, these life-saving devices will become the global standard of care—protecting patients, healthcare workers, and communities worldwide.

For more information about implementing auto-disable syringes in your healthcare facility or distribution network, visit our comprehensive product catalog or contact our technical team for customized solutions.