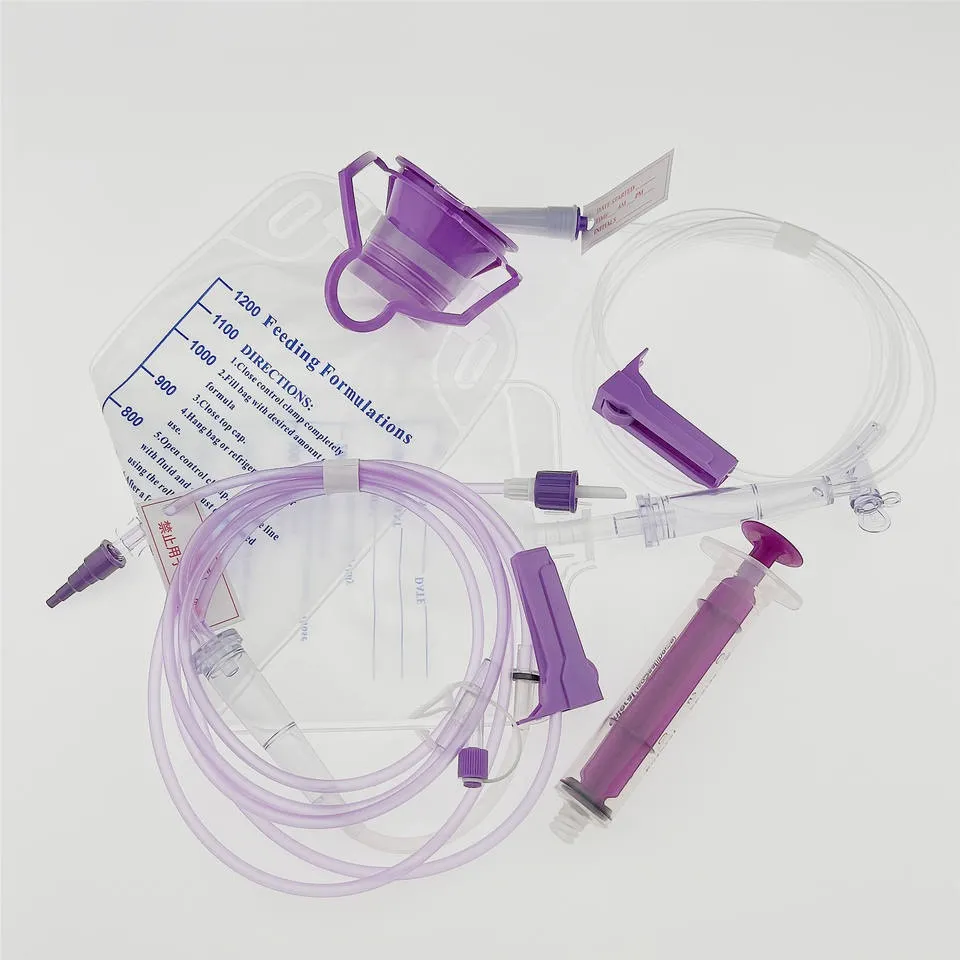
In modern healthcare, enteral nutrition has become a cornerstone of patient care, supporting millions of individuals who cannot consume food orally due to medical conditions, surgical procedures, or chronic illnesses. At the heart of this essential therapy lies a seemingly simple yet critically important device: the Enteral Syringe. However, the journey from basic feeding tubes to today’s sophisticated ENFit-compliant systems represents a profound shift in medical device design philosophy—one driven by tragic preventable errors and an unwavering commitment to patient safety.
This article explores the market landscape, clinical applications, development rationale, and future directions of enteral syringes, providing procurement professionals and healthcare administrators with comprehensive insights into this essential medical device category.
The Development Genesis: Solving a Fatal Problem
The Misconnection Crisis
The primary catalyst behind modern Enteral Syringes development was a sobering reality: medical tubing misconnections were causing serious patient harm and preventable deaths. Before standardization, enteral feeding syringes used luer connectors—the same interface found on intravenous (IV) systems. This compatibility created dangerous scenarios where nutrition formulas could accidentally be administered intravenously, or conversely, where IV medications could enter the gastrointestinal tract.
Clinical literature documented numerous cases of:
- Enteral feeding solutions mistakenly connected to central venous catheters, causing fat embolisms and sepsis
- Intravenous medications inadvertently delivered through feeding tubes, resulting in therapeutic failures
- Fatal air embolisms when feeding systems were connected to IV lines
These incidents, while individually rare, collectively represented a systemic design flaw that demanded industry-wide correction.

The ISO 80369-3 Solution
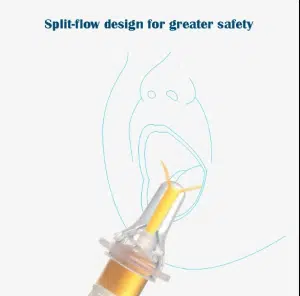
In response, the International Organization for Standardization (ISO) developed the 80369-3 standard, introducing the ENFit connector—a unique interface designed exclusively for enteral applications. This innovation fundamentally changed the Enteral Syringes manufacturing landscape, establishing a new baseline for product development focused on incompatibility by design.
The ENFit system’s core principle: enteral devices should be physically incapable of connecting to IV, respiratory, or other non-enteral systems. This “forcing function” approach to safety represents a paradigm shift from relying on user vigilance to embedding safety into the device architecture itself.
Market Applications: Diverse Clinical Scenarios
Hospital & Acute Care Settings
Enteral Syringes serve critical functions across multiple hospital departments:
Intensive Care Units (ICUs) rely heavily on enteral nutrition for mechanically ventilated patients unable to maintain oral intake. Syringes ranging from 20ml to 60ml facilitate medication administration and nutritional bolus feeding, while larger 100ml syringes support continuous feeding protocols.
Surgical Recovery Units use enteral syringes for post-operative patients, particularly following gastrointestinal, neurological, or maxillofacial procedures. The ability to deliver precise medication doses through nasogastric or gastrostomy tubes is essential for pain management and antibiotic prophylaxis when oral administration isn’t feasible.
Pediatric Departments require the full spectrum of syringe sizes, from 1ml for neonatal medication delivery to larger volumes for older children. The latex-free, DEHP-free materials address pediatric populations’ heightened sensitivity to allergens and chemical exposure.
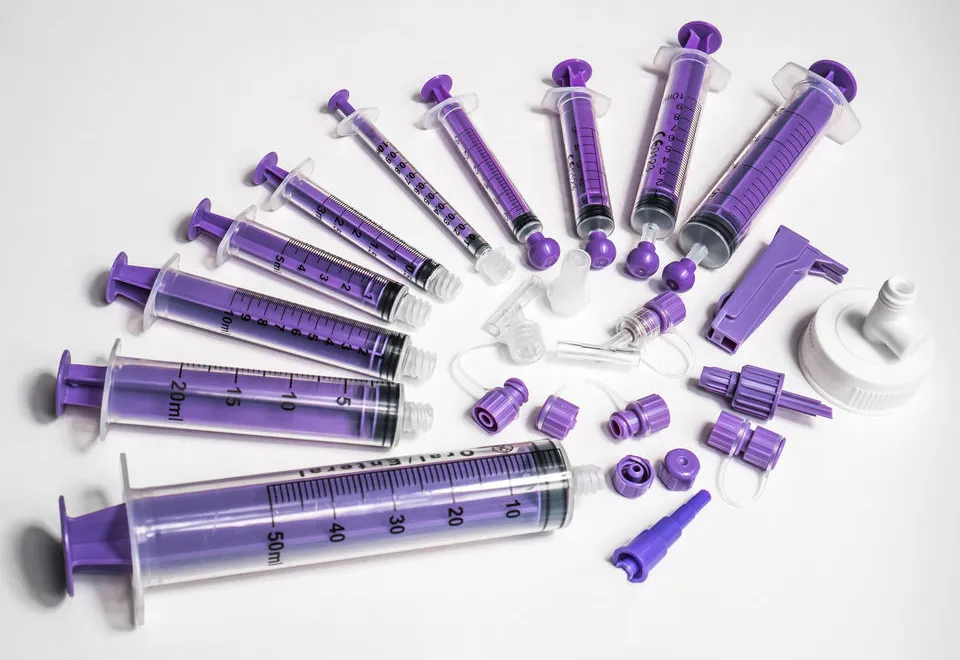
Long-Term Care & Rehabilitation Facilities
Nursing homes and rehabilitation centers represent substantial market segments for custom Enteral Syringes. Residents with neurological conditions (stroke, Parkinson’s disease, dementia) often require long-term enteral support. The thumb control ring design becomes particularly valuable in these settings, where caregivers frequently manage multiple patients and benefit from one-handed administration capabilities.
Home Healthcare: The Growing Frontier
Perhaps the most rapidly expanding application area is home-based enteral nutrition. Advances in medical technology and healthcare economics have shifted many long-term enteral feeding patients from institutional settings to home care. This transition creates unique requirements:
- User-friendly design for non-professional caregivers
- Safety features that prevent errors in less controlled environments
- Durable packaging that withstands home storage conditions
- Clear markings for volume measurement by family members
The market for home healthcare enteral products is projected to grow at 7.2% CAGR through 2030, driven by aging populations and preference for home-based care models.
Functional Advantages: Engineering for Excellence
Material Science Innovations
Modern Enteral Syringes incorporate advanced materials that address historical complications:
DEHP-Free Formulations: Di(2-ethylhexyl) phthalate, previously common in medical plastics, has been eliminated due to concerns about endocrine disruption and reproductive toxicity. Alternative plasticizers maintain flexibility and durability without these risks.
Latex-Free Synthetic Rubber Plungers: Natural rubber latex allergies affect approximately 1-6% of the general population and up to 12% of healthcare workers. Synthetic rubber plungers eliminate this risk while maintaining the seal integrity and smooth operation essential for accurate administration.
Low Dead Space Design: In smaller capacity syringes (1ml, 3ml, 5ml), minimizing residual volume in the connector becomes critical. The male protrusion within the ENFit female connector reduces dead space from approximately 0.3ml to less than 0.1ml—a significant improvement when administering expensive medications or precise pediatric doses.

Ergonomic Refinements
The thumb control ring represents thoughtful human factors engineering. Healthcare workers administering multiple doses throughout a shift experience reduced hand fatigue and improved dosing accuracy. This seemingly minor feature has substantial implications for workflow efficiency and occupational health.
Precision-crafted O-ring plungers ensure consistent resistance throughout the administration stroke, providing tactile feedback that helps clinicians detect tube occlusions or positioning problems before complications arise.
Current Limitations & Areas for Improvement
Despite significant advances, Enteral Syringes face several challenges that represent opportunities for innovation:
Material Sustainability Concerns
Current manufacturing predominantly relies on single-use plastics, contributing to healthcare’s substantial environmental footprint. While sterility and safety requirements justify disposable designs, the industry lacks economically viable recycling programs for used enteral devices. Biodegradable polymers that maintain necessary performance characteristics remain in early development stages.
Cost Barriers in Resource-Limited Settings
ENFit-compliant Enteral Syringes typically cost 30-50% more than legacy luer-based products. While economically developed healthcare systems have largely absorbed this transition, hospitals in lower-income regions continue using older, less safe alternatives. Manufacturers face the challenge of maintaining safety standards while developing cost-effective solutions for global markets.
Compatibility Transition Challenges
The healthcare industry’s migration to ENFit standards created a transitional period where facilities maintain inventory of both legacy and compliant devices. This dual-system approach introduces its own risks and inventory management complexities. Complete system conversion requires coordinated replacement of feeding tubes, syringes, administration sets, and related accessories—a significant capital expenditure many facilities struggle to prioritize.
Limited Smart Technology Integration
Unlike some medical devices that have incorporated digital connectivity and smart features, Enteral Syringes remain purely mechanical. Opportunities exist for innovation in:
- Dose tracking and electronic health record integration
- Volume verification systems
- Temperature-sensitive indicators for refrigerated formulas
- RFID tags for inventory management and recall traceability
However, adding electronic components increases complexity, cost, and potential failure points—factors that must be carefully balanced against clinical benefits.

Market Trends & Future Directions
Customization & Private Labeling Growth
The rise of hospital group purchasing organizations and integrated healthcare networks has accelerated demand for custom Enteral Syringes with institutional branding. Private label products strengthen supply chain relationships and can include facility-specific features like color coding for different formula types or departmental protocols.
Leading Enteral Syringes manufacturers now offer comprehensive customization services including package design, volume calibration modifications, and co-development of specialized features for unique clinical applications.
Regulatory Landscape Evolution
Global harmonization of medical device standards continues to shape market dynamics. The European Union’s Medical Device Regulation (MDR) and similar frameworks in other regions impose stricter quality management and post-market surveillance requirements. Manufacturers demonstrating robust compliance capabilities gain competitive advantages in procurement decisions.
Emerging Market Expansion
Asia-Pacific healthcare infrastructure development, Middle Eastern medical tourism growth, and Latin American healthcare modernization initiatives represent substantial expansion opportunities. However, success in these markets requires navigating diverse regulatory environments, price sensitivity, and varying clinical practice patterns.
Value-Based Procurement Shifts
Healthcare purchasers increasingly evaluate medical devices through total cost of ownership rather than unit price alone. Enteral Syringes that reduce complication rates, decrease nursing time, or minimize medication waste deliver value that justifies premium pricing. Manufacturers who provide compelling health economics data alongside product specifications position themselves advantageously in competitive tenders.


Procurement Considerations for Healthcare Organizations
Quality Verification Beyond Price
When evaluating Enteral Syringes suppliers, procurement professionals should assess:
Manufacturing Standards: ISO 13485 certification, clean room classifications, and quality management system maturity indicate production reliability.
Testing Protocols: Comprehensive verification including dimensional accuracy, leak testing, plunger force consistency, and sterility assurance demonstrates commitment to quality.
Supply Chain Resilience: Recent global disruptions highlighted the importance of diversified sourcing, inventory buffering, and supplier financial stability.
Regulatory Documentation: Complete technical files, certificates of conformity, and biocompatibility testing support institutional compliance obligations.
Balancing Standardization with Clinical Needs
While product consolidation reduces inventory complexity and costs, clinical departments may have legitimate requirements for specific features. Successful procurement strategies involve multidisciplinary stakeholders—clinicians, pharmacy, infection control, and supply chain—in specification development.
Sustainability Initiatives
Progressive healthcare organizations increasingly prioritize environmental considerations in procurement decisions. Engaging Enteral Syringes manufacturers in discussions about packaging reduction, recycled content, and take-back programs can drive industry-wide improvements.

Conclusion: A Device Category Transformed by Safety Imperatives
The modern Enteral Syringe represents far more than a simple plastic tube and plunger. It embodies a fundamental reconceptualization of medical device design philosophy—one that prioritizes inherent safety over user-dependent precautions. The development journey from basic feeding syringes to ISO 80369-3 compliant ENFit systems demonstrates healthcare’s capacity for systemic improvement when confronting preventable harm.
For procurement professionals, understanding this evolution provides context for evaluating suppliers, justifying costs, and advocating for quality standards. The slight premium paid for ENFit-compliant, material-safe, ergonomically optimized Enteral Syringes represents an investment in patient safety and clinical efficiency that delivers returns through reduced complications, improved workflow, and enhanced institutional reputation.
As the market continues evolving—driven by regulatory harmonization, technological innovation, sustainability imperatives, and global healthcare access expansion—organizations that partner with forward-thinking Enteral Syringes manufacturers position themselves to benefit from ongoing improvements while maintaining the safety standards that patients deserve and regulations demand.
The enteral syringe’s story is ultimately one of healthcare’s perpetual journey toward safer, more effective care delivery. In this context, even the humblest medical device carries profound importance—and deserves the thoughtful evaluation and strategic procurement that supports optimal patient outcomes.