In modern healthcare, patient safety and clinical efficiency go hand-in-hand. Among the most critical innovations in medical devices are safety pen needles – precision-engineered tools that have revolutionized intravenous access while dramatically reducing needlestick injuries among healthcare workers. This comprehensive guide explores the development, manufacturing landscape, and key considerations for selecting a reliable pen needles manufacturer.
What Are Safety Pen Needles?
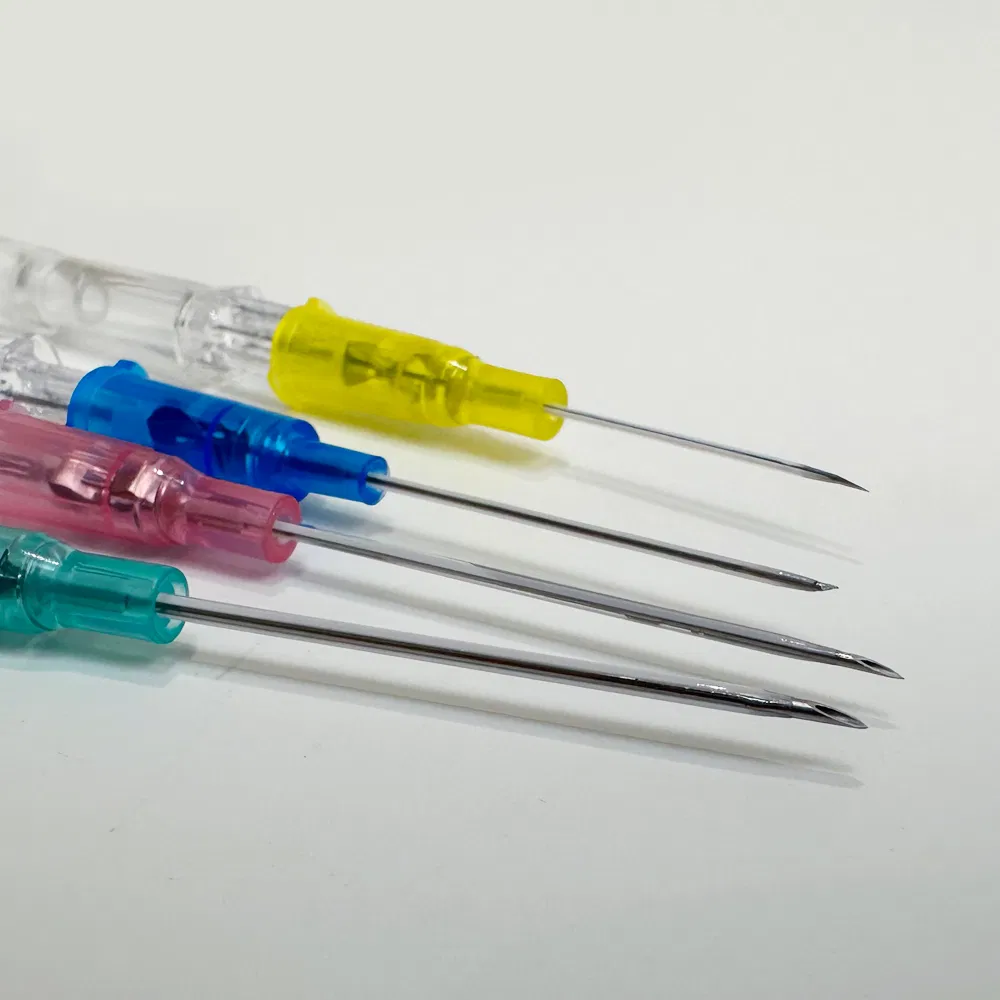
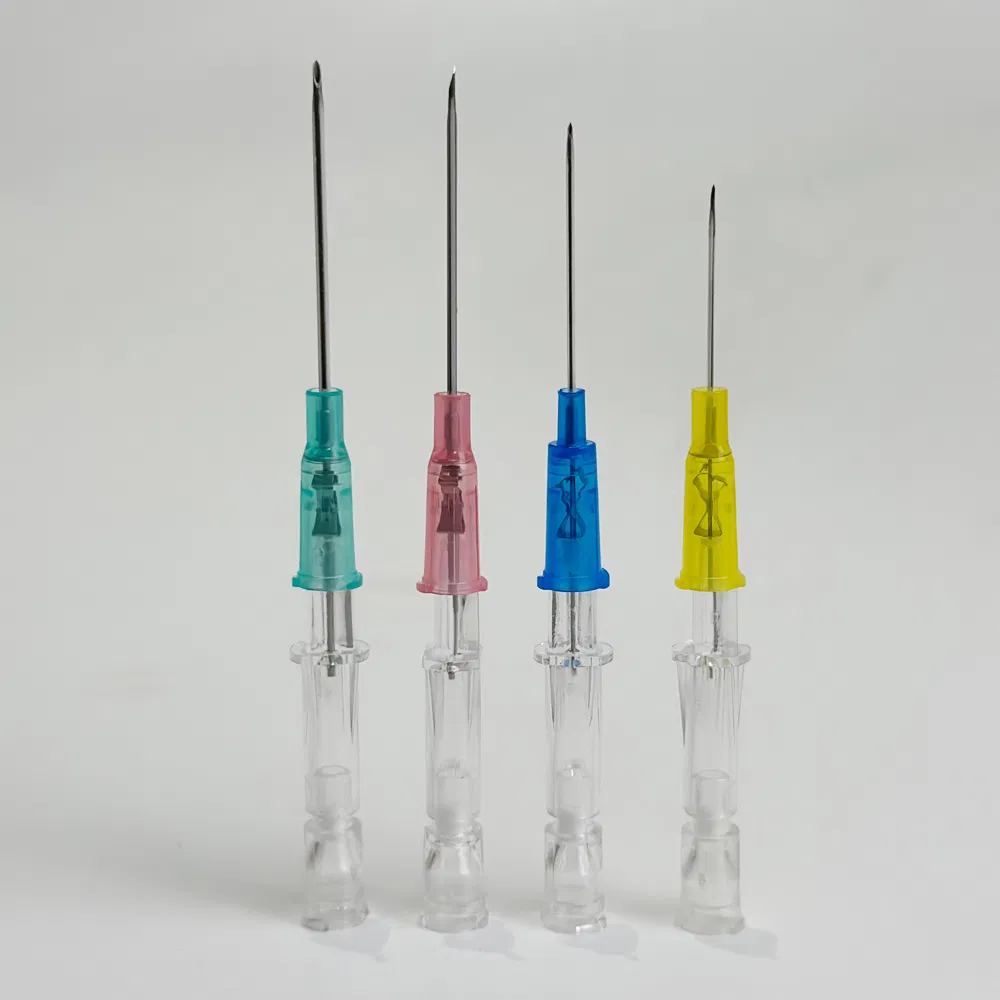
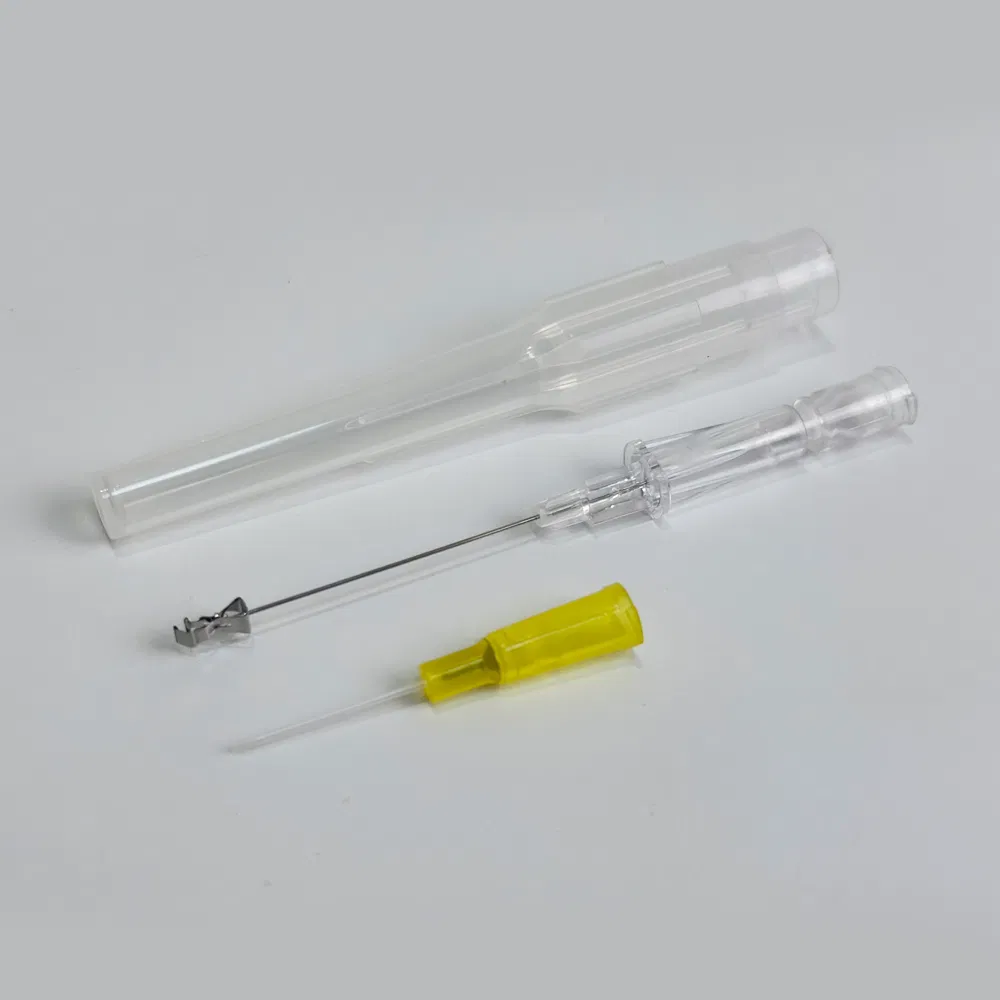
Safety pen needles, also known as winged infusion sets or butterfly needles, are specialized medical devices designed for venipuncture and short-term intravenous therapy. Unlike traditional straight needles, these devices feature:
- Flexible stabilization wings that provide superior control during insertion
- Retractable safety mechanisms that automatically shield the needle post-procedure
- Transparent flashback chambers for immediate visual confirmation of venous access
- Hypoallergenic materials suitable for patients with latex sensitivities
The “pen-grip” design allows clinicians to hold the device similar to writing with a pen, offering unprecedented precision and comfort during delicate procedures.
The Evolution of Safety Pen Needles: A Historical Perspective
Early Development (1950s-1970s)
The concept of winged infusion devices emerged in the 1950s when healthcare professionals recognized the need for more stable venous access, particularly in pediatric and geriatric populations. The original butterfly needles featured simple plastic wings but lacked safety mechanisms, leaving healthcare workers vulnerable to accidental needlestick injuries.
The Safety Revolution (1980s-1990s)
The HIV/AIDS epidemic of the 1980s brought needlestick safety into sharp focus. Studies revealed that healthcare workers faced significant occupational exposure risks, prompting regulatory bodies worldwide to mandate safety-engineered devices. This period saw the introduction of the first retractable needle mechanisms and passive safety features.
Modern Innovations (2000s-Present)
Today’s safety pen needles represent the culmination of decades of research and development. Advanced manufacturing techniques have enabled:
- Ultra-sharp tri-beveled needle tips with silicone coating for pain-free insertion
- Automatic retraction systems that activate with a single click
- Color-coded hubs for quick gauge identification
- Ergonomic wing designs that accommodate various hand sizes and insertion angles
- Enhanced transparency in flashback chambers for improved visibility
The global push toward zero needlestick injuries has made safety pen needles the standard of care in hospitals, infusion centers, and home healthcare settings worldwide.
Global Manufacturing Landscape: Where Safety Pen Needles Are Made
Major Manufacturing Hubs
Asia-Pacific Region
China, particularly the Jiangsu and Guangdong provinces, has emerged as the world’s largest production center for medical needles and IV catheters. The region benefits from:
- Established supply chains for medical-grade polymers and stainless steel
- Advanced automation and quality control infrastructure
- Cost-effective production with ISO 13485 certification capabilities
- Proximity to raw material sources and global shipping routes
Europe
Germany, France, and Switzerland maintain premium manufacturing facilities focused on:
- High-precision needle grinding and beveling technology
- Strict compliance with EU Medical Device Regulation (MDR)
- Innovation in safety mechanism designs
- Small-batch specialty products for niche applications
North America
The United States hosts several specialized manufacturers emphasizing:
- FDA-registered facilities with cGMP compliance
- Rapid prototyping and custom OEM development
- Domestic supply chain security for critical healthcare infrastructure
Manufacturing Standards Across Regions
Regardless of location, reputable safety needle suppliers adhere to international standards including:
- ISO 13485: Quality management systems for medical devices
- ISO 23908: Sharps injury protection requirements
- ASTM F2624: Standard test methods for needlestick injury prevention
- EN ISO 7886: Sterile hypodermic needles for single use

Key Advantages of Working with Established Pen Needles Manufacturers
1. Quality Consistency Through Process Control
Leading manufacturers implement rigorous quality assurance protocols:
Raw Material Selection: Medical-grade PVC tubing undergoes biocompatibility testing per ISO 10993 standards. Stainless steel needles (typically 304 or 316L grade) are inspected for metallurgical composition and surface defects.
Precision Manufacturing: Automated needle grinding achieves tolerances within ±0.01mm. Tri-beveling processes create optimal penetration angles (typically 12-15 degrees) for minimal tissue trauma.
Sterilization Validation: Ethylene oxide (EO) sterilization cycles are validated to achieve a Sterility Assurance Level (SAL) of 10⁻⁶, meaning less than one in a million chance of non-sterility.
In-Process Testing: Every production batch undergoes:
- Needle sharpness testing using penetration force meters
- Flashback chamber clarity verification
- Retraction mechanism functionality checks (100% inspection on critical safety features)
- Packaging integrity testing through dye penetration methods
2. Economies of Scale
High-volume manufacturers produce millions of units monthly, enabling:
- Lower per-unit costs through optimized material utilization and automated assembly
- Inventory availability to meet sudden demand spikes during healthcare emergencies
- Investment in R&D for continuous product improvement without passing development costs to buyers
3. Technical Expertise and Support
Experienced pen needles manufacturers offer value beyond the product itself:
- Clinical training programs for proper insertion techniques and safety mechanism activation
- Product selection guidance based on specific patient populations and clinical applications
- Custom development capabilities for specialized requirements (extended tubing lengths, unique connector types, proprietary safety designs)
- Regulatory documentation support including technical files, safety data sheets, and biocompatibility reports
4. Regulatory Compliance Infrastructure
Established manufacturers maintain:
- Dedicated regulatory affairs teams tracking changing requirements across global markets
- Post-market surveillance systems to monitor device performance and adverse events
- Traceability systems using lot coding and serialization for rapid recall capability if needed
- Regular external audits by notified bodies (EU), FDA inspectors, or other regulatory authorities
5. Supply Chain Resilience
Professional manufacturers build redundancy into their operations:
- Multiple sourcing strategies for critical components to prevent single-point failures
- Safety stock protocols maintaining 30-90 days of finished goods inventory
- Geographic diversification with backup production facilities
- Long-term material contracts protecting against price volatility and shortages
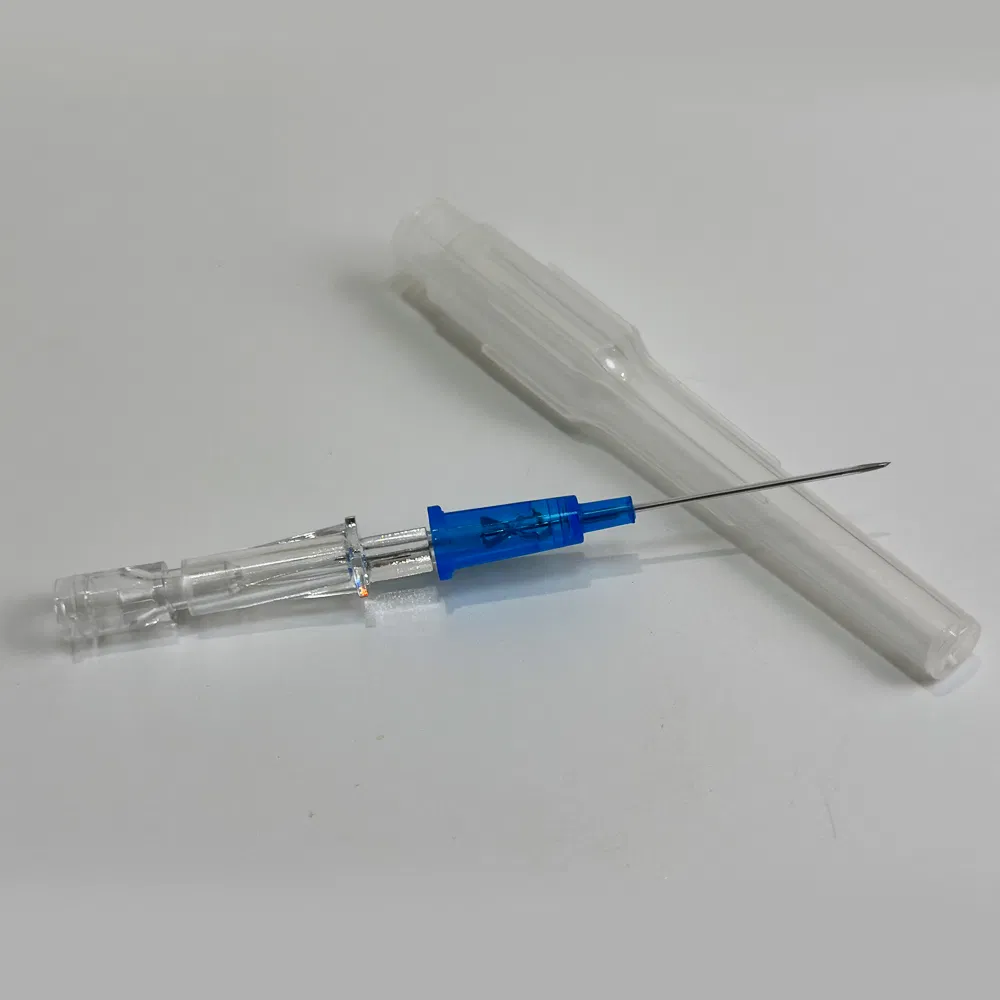
How Safety Pen Needles Are Manufactured: Inside the Production Process
Understanding the manufacturing workflow helps buyers appreciate quality differentials:
Step 1: Needle Production
Stainless steel wire is cut to precise lengths, then undergoes:
- Point grinding: Diamond wheels create the tri-bevel tip geometry
- Siliconization: A microscopic coating reduces friction during insertion
- Laser marking: Gauge numbers and manufacturer codes are etched
- Vision inspection: Automated cameras detect burrs or irregularities
Step 2: Catheter Assembly
Medical-grade PVC tubing is:
- Cut and formed: Precision cutting ensures consistent lengths
- Hub attachment: Wings and connector hubs are ultrasonically welded or adhesive-bonded
- Needle insertion: Needles are press-fit into catheter hubs with controlled force
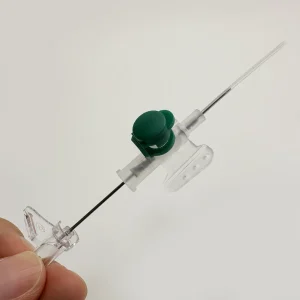
Step 3: Safety Mechanism Integration
The retractable system installation involves:
- Spring loading: Compression springs are precisely calibrated for reliable activation
- Shielding components: Plastic shields are aligned to cover the full needle length
- Activation testing: Each unit’s retraction mechanism is tested for smooth, complete needle withdrawal
Step 4: Final Assembly and Packaging
- Tubing attachment: Extension tubing with luer connectors is secured
- Visual inspection: Operators check for proper assembly and cosmetic defects
- Blister sealing: Devices are placed in thermoformed trays and sealed with medical-grade Tyvek
- Outer packaging: Units are boxed with printed instructions for use (IFU)
Step 5: Sterilization
Pallets of packaged devices enter:
- EO chambers: Controlled temperature, humidity, and gas concentration for 12-24 hours
- Aeration rooms: Residual EO is removed to safe levels per ISO 10993-7
- Quarantine: Products remain in controlled storage until sterility validation is complete
Step 6: Quality Release
Before shipment, statistical sampling includes:
- Sterility testing (USP <71>): Samples are incubated in growth media to confirm no microbial contamination
- Physical testing: Needle sharpness, retraction force, and flow rate measurements
- Packaging integrity: Seal strength testing and accelerated aging studies
- Certificate of Analysis: Each lot receives documentation verifying compliance with specifications

Product Features That Define Quality in Safety Pen Needles
When evaluating different needles from various manufacturers, discerning buyers focus on these critical attributes:
Needle Design Specifications
Ultra-Sharp Tri-Bevel Geometry: Premium manufacturers use computer-controlled grinding to create three precisely angled cutting edges. This design slices through skin rather than tearing tissue, resulting in:
- Reduced insertion force (typically 20-30% less than conventional bevels)
- Minimal pain perception reported by patients
- Lower risk of vein wall damage and hematoma formation
Siliconization Quality: Not all silicone coatings are equal. Superior coatings:
- Maintain lubricity through multiple insertions (important if repositioning is needed)
- Don’t flake or create particles that could enter the bloodstream
- Remain stable across temperature variations during shipping and storage
Gauge and Length Options: Professional product lines offer comprehensive ranges:
- 18G-20G (large bore): Rapid fluid resuscitation, blood product administration
- 22G (standard): General IV therapy, medication infusion
- 24G-26G (small bore): Pediatric patients, fragile veins, subcutaneous access
Safety Mechanism Performance
Activation Method:
- Automatic/Passive: Retracts without user action when removed from vein (ideal for high-stress emergency situations)
- Semi-automatic: Requires single-handed button press or thumb slide (provides controlled activation timing)
Audible/Tactile Confirmation: Quality devices provide clear feedback when the safety mechanism engages – a click sound or resistance change confirming full needle shielding.
Lockout Features: After activation, the mechanism should permanently disable, preventing needle re-exposure even under intentional manipulation.
Winged Stabilization Design
Wing Flexibility: Medical-grade thermoplastic elastomers (TPE) provide:
- Enough rigidity for secure gripping during insertion
- Sufficient flexibility to fold flat for comfortable taping against skin
- Memory to return to original shape if repositioning is needed
Wing Size Optimization: Typical dimensions range from 15-25mm wingspan. Larger wings offer more control but may be cumbersome for small insertion sites; smaller wings reduce bulk but require more precise handling.
Texture and Grip: Micro-textured surfaces or ribbed patterns prevent slippage from gloved fingers, especially when wet with antiseptic solutions or blood.
Flashback Chamber Transparency
Material Selection: Crystal-clear polycarbonate or PETG plastics ensure:
- Immediate visibility of blood return (flashback) confirming venous access
- No color distortion that could mask subtle blood flow patterns
- Resistance to scratching or clouding during handling
Chamber Volume: Optimal designs balance rapid flashback visualization (smaller volume fills faster) with sufficient capacity to maintain visible blood during securing procedures.
Hypoallergenic Material Composition
Latex-Free Certification: Complete absence of natural rubber latex eliminates Type I hypersensitivity risks for sensitized patients and healthcare workers.
DEHP-Free PVC Options: Advanced manufacturers offer alternatives to di(2-ethylhexyl) phthalate plasticizers, addressing concerns about reproductive and developmental toxicity.
Biocompatibility Testing: Full ISO 10993 evaluation confirms:
- No cytotoxic effects on living cells
- Minimal inflammatory response during short-term contact
- Absence of sensitization potential through repeated exposure
How to Select the Right Safety Pen Needle Supplier: A Buyer’s Decision Framework
Choosing among numerous manufacturers requires systematic evaluation across multiple dimensions:
1. Regulatory Credentials and Market Access
Certification Portfolio: Verify the manufacturer holds relevant approvals for your target markets:
- FDA Registration (USA): Establishment registration and device listing in 510(k) database
- CE Marking (Europe): Compliance with EU MDR 2017/745, issued by a notified body
- NMPA Approval (China): Registration for Class II medical devices
- TGA Registration (Australia), Health Canada License, ANVISA (Brazil), etc.
Audit Trail: Request evidence of recent third-party audits:
- ISO 13485 certification from accredited bodies (TÜV, BSI, SGS)
- Unannounced FDA inspections with no significant observations (Form 483)
- Customer quality audits from major hospital systems or GPOs
Warning Letter History: Search FDA’s public database and EU NANDO for compliance issues. A clean record indicates consistent adherence to quality standards.
2. Manufacturing Capacity and Scalability
Production Volume: Align supplier capacity with your demand profile:
- Startup distributors: 50,000-500,000 units/quarter may need flexible MOQ suppliers
- Regional distributors: 1-5 million units/quarter require stable mid-tier manufacturers
- National GPOs: 10+ million units/quarter demand top-tier producers with multiple production lines
Lead Time Reliability: Request on-time delivery metrics (OTIF – On Time In Full):
- Industry standard: >95% OTIF performance
- Investigate backup plans for material shortages or equipment breakdowns
Surge Capacity: During pandemic or emergency situations, can the manufacturer scale production by 50-150% within 30-60 days?
3. Product Range and Customization Capabilities
Standard SKU Breadth: Comprehensive manufacturers offer:
- Multiple gauge options (18G, 20G, 22G, 24G, 26G)
- Varied needle lengths (19mm, 25mm, 32mm standard; custom lengths available)
- Different tubing configurations (short 7-inch, standard 12-inch, extended 15-inch)
- Safety mechanism variations (automatic vs. semi-automatic activation)
OEM/ODM Services: For private-label or custom products, evaluate:
- Minimum order quantities: Can they accommodate 10,000-unit pilots or require 100,000+ commitments?
- Design modification scope: Will they alter wing shapes, add proprietary safety features, or create unique packaging?
- Intellectual property protection: Do they sign NDAs and respect your design ownership?
- Timeline for customization: Typical development cycles range 3-6 months from concept to production
Color Coding Options: Some buyers prefer standardized international color schemes (pink=20G, blue=22G, yellow=24G), while others want custom colors for brand differentiation.
4. Quality Systems and Testing Protocols
In-Process Quality Control: Tour the facility (virtually or in-person) to observe:
- Statistical process control (SPC) implementation on critical parameters
- Automated inspection systems (vision cameras, force sensors)
- Environmental monitoring in production areas (particle counts, microbial sampling)
- Operator training programs and competency verification
Finished Product Testing: Request the manufacturer’s standard test plan:
- Needle sharpness: Penetration force testing per ISO 7864
- Safety mechanism reliability: Activation force and retraction completeness (sample size: typically 13 units per ISO 23908)
- Sterility assurance: USP <71> testing frequency (per lot or statistically sampled)
- Packaging integrity: Seal strength and dye penetration testing
Traceability Systems: Ensure full lot traceability from raw materials to finished product:
- Raw material certificates of analysis (COA) retained
- Production batch records with operator identification and equipment logs
- Serialization capabilities for unit-level tracking if required by regulations
5. Pricing Structure and Total Cost of Ownership
Unit Price Analysis: Don’t focus solely on per-unit cost. Consider:
- Volume tiering: How much do prices decrease at 100K, 500K, 1M unit thresholds?
- Payment terms: Net 30, 60, or 90 days? Letter of credit requirements?
- Currency fluctuation: Fixed USD/EUR pricing or tied to exchange rates?
Hidden Costs: Factor in the complete cost picture:
- Freight and logistics: FOB, CIF, or DDP terms? Customs duties and import taxes?
- Inventory carrying costs: Can the supplier hold safety stock or dropship directly to customers?
- Quality failures: What are the costs of recalls, returns, or customer complaints from defective products?
- Regulatory maintenance: Does the supplier cover recertification fees or pass costs to buyers?
Value-Added Services: Premium suppliers may justify higher unit prices through:
- Consignment inventory programs
- Just-in-time delivery reducing your warehouse needs
- Technical training for your sales team or end-customers
- Co-marketing support (product photography, technical brochures, clinical studies)
6. Technical Support and Responsiveness
Pre-Sales Engagement: Evaluate supplier responsiveness during the inquiry phase:
- How quickly do they respond to initial contact? (24-48 hours is standard)
- Do they ask probing questions to understand your specific needs, or push generic products?
- Can they provide detailed technical specifications, CAD drawings, or samples promptly?
Post-Sales Support: Investigate ongoing support quality:
- Complaint handling: What’s the response time for quality issues? Do they conduct root cause investigations?
- Technical documentation: Are instructions for use (IFU) available in multiple languages?
- Field actions: How do they manage recalls or field safety notices if required?
Account Management: Will you have a dedicated contact person, or route through general customer service? For larger accounts, dedicated account managers provide:
- Proactive communication about production schedules
- Early notification of raw material shortages or price changes
- Quarterly business reviews to optimize product mix and inventory
7. Sustainability and Ethical Manufacturing Practices
Environmental Stewardship: Increasingly, healthcare organizations prioritize suppliers with:
- ISO 14001 environmental management certification
- Waste reduction programs: Recycling of production scrap, solvent recovery systems
- Energy efficiency: Solar installations, LED lighting, optimized HVAC systems
- Sustainable packaging: Reduced plastic use, recycled content in outer cartons
Social Responsibility: Verify ethical labor practices through:
- SMETA/Sedex audits: Evaluating working conditions, fair wages, and child labor policies
- Conflict minerals compliance: Certification that tantalum, tin, tungsten, and gold (3TG) are ethically sourced
- Corporate social responsibility reports: Transparency about community impact and employee welfare programs
8. Risk Mitigation and Business Continuity
Financial Stability: Assess the manufacturer’s long-term viability:
- Years in business (established manufacturers with 10+ years demonstrate resilience)
- Financial disclosures if publicly traded, or Dun & Bradstreet credit reports
- Insurance coverage (product liability, recall insurance, business interruption)
Geographic Diversification: Single-site manufacturers pose supply chain risks. Prefer suppliers with:
- Multiple production facilities in different regions
- Qualified backup suppliers for critical components
- Business continuity plans tested through tabletop exercises
Intellectual Property Protection: For custom designs:
- Sign non-disclosure agreements (NDAs) before sharing specifications
- Clearly define IP ownership in contracts (your design vs. their manufacturing process)
- Include non-compete clauses preventing them from selling your custom design to competitors

Application-Specific Considerations: Matching Products to Clinical Needs
Different healthcare settings demand tailored safety pen needle specifications:
Pediatric Applications
Design Priorities:
- Smaller gauges (24G-26G) minimize pain and vessel trauma in small veins
- Shorter needle lengths (19mm) reduce risk of through-and-through punctures
- Vibrant colors (pediatric-friendly packaging) reduce patient anxiety
- Reduced insertion force enables gentle technique for fragile vessels
Clinical Evidence: Studies show that 25G safety pen needles achieve 92% first-stick success rates in pediatric populations compared to 78% with standard butterfly needles, reducing procedural stress for both patient and clinician.
Geriatric and Oncology Settings
Design Priorities:
- Ultra-sharp bevels penetrate fragile, sclerotic veins with minimal trauma
- Enhanced wing grip accommodates hand tremors or reduced dexterity
- Extended dwell time materials withstand longer infusion periods
- Transparent hubs allow monitoring for infiltration or phlebitis
Clinical Benefits: For chemotherapy patients with compromised peripheral access, safety pen needles extend the usable lifespan of remaining veins by reducing scarring from traumatic insertions.
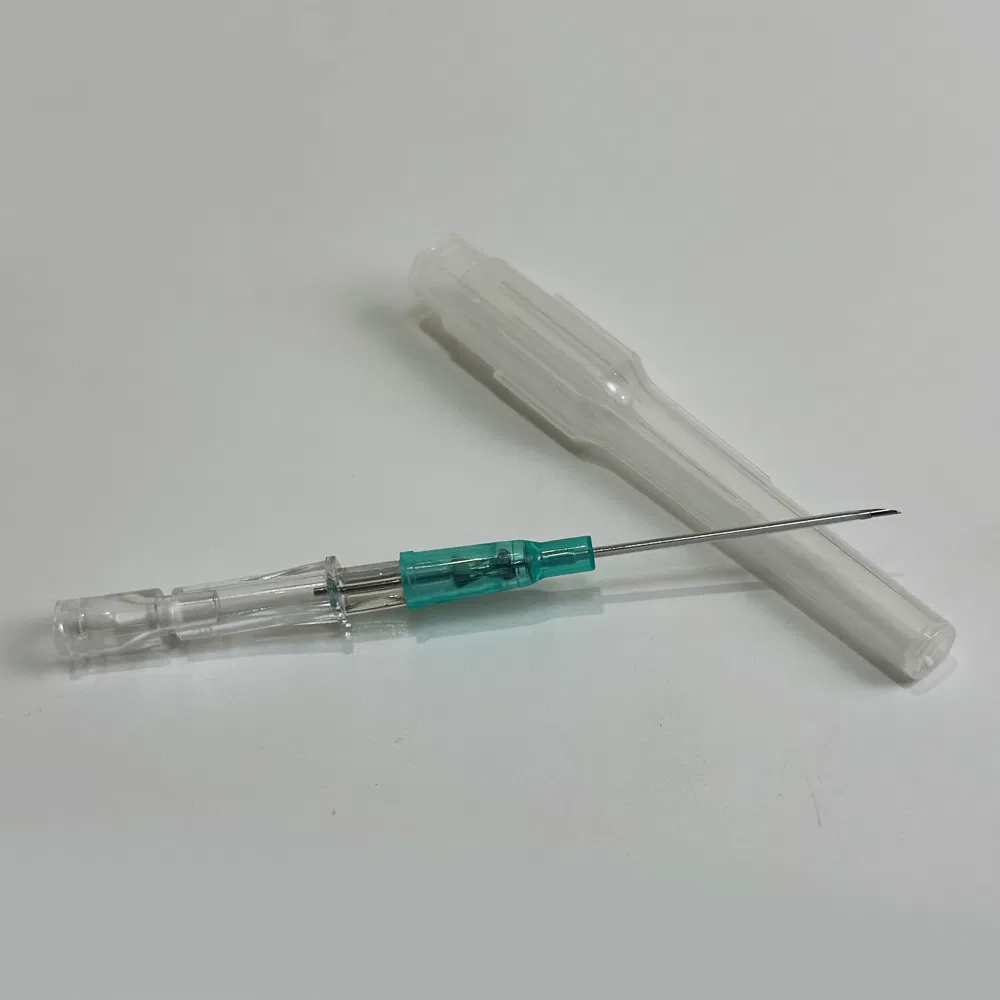

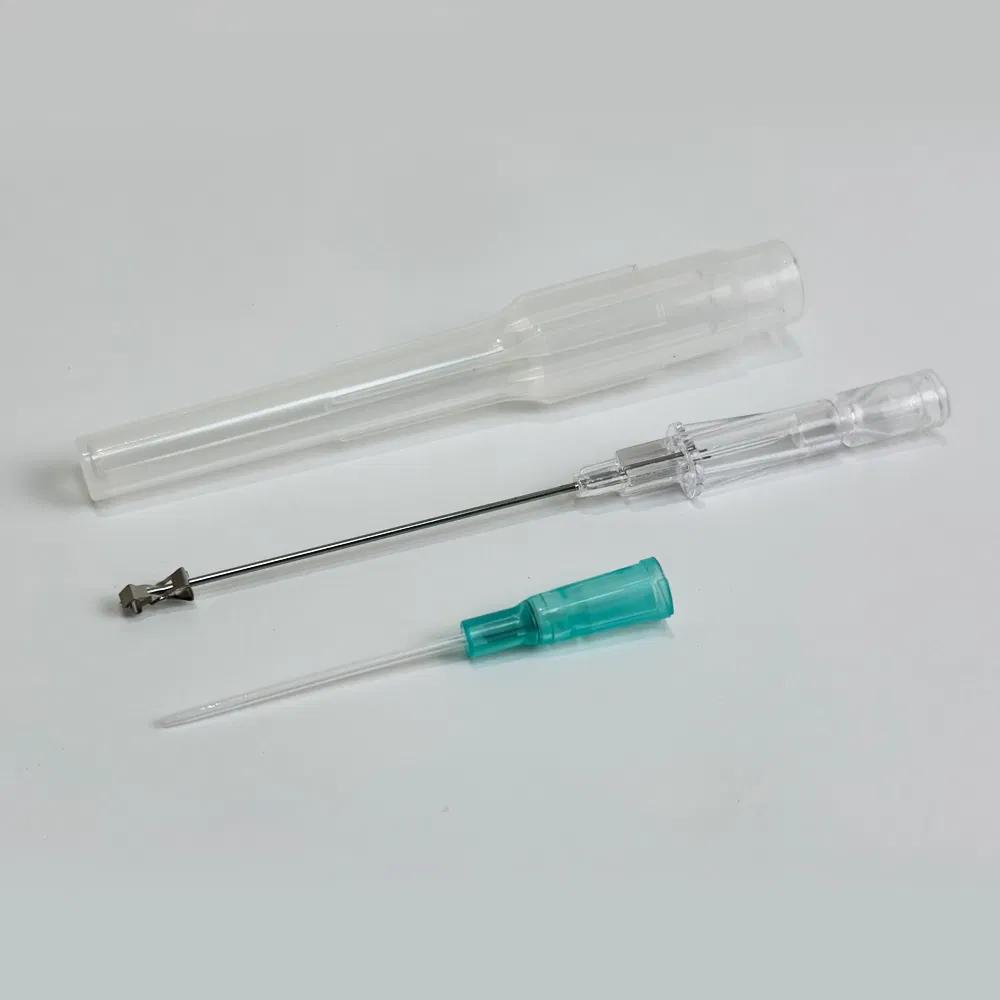
Emergency and Pre-Hospital Care
Design Priorities:
- Larger gauges (18G-20G) enable rapid fluid resuscitation
- Automatic safety activation eliminates user steps during high-stress situations
- Robust construction withstands rough handling in ambulances or field environments
- Clear packaging allows rapid gauge identification without reading fine print
Performance Metrics: Emergency departments report 30% reduction in needlestick injuries after transitioning to automatic-retraction safety pen needles versus manual safety devices requiring conscious activation.
Home Healthcare and Self-Administration
Design Priorities:
- Intuitive safety activation suitable for patients with limited medical training
- Comfortable wings for self-insertion at awkward angles
- Clear visual indicators confirming successful venous access
- Secure fixation preventing dislodgement during activities of daily living
Patient Outcomes: Home infusion therapy patients using safety pen needles report 85% satisfaction scores compared to 68% with standard IV catheters, citing easier self-insertion and greater confidence in secure placement.
Future Trends in Safety Pen Needle Technology
The industry continues to innovate, with emerging developments including:
Smart Connected Devices
Integration with IoT Platforms: Next-generation devices may incorporate:
- RFID tags for automated inventory tracking and expiration management
- NFC chips linking to digital instructions for use and training videos
- Insertion sensors providing real-time feedback on angle and depth (training applications)
Advanced Materials Science
Biocompatible Coatings: Research explores:
- Antimicrobial surfaces reducing catheter-related bloodstream infections (CRBSI)
- Hydrophilic polymers further reducing insertion friction and patient discomfort
- Biodegradable components for environmentally sustainable medical waste management
Personalized Medicine Applications
Patient-Specific Customization: 3D printing technologies may enable:
- Custom needle lengths based on ultrasound measurements of vein depth
- Personalized wing geometries accommodating individual hand sizes and grip preferences
- On-demand manufacturing reducing inventory costs and waste
Artificial Intelligence Integration
Procedural Guidance Systems: AI-powered devices could offer:
- Vein visualization using integrated near-infrared imaging
- Predictive success modeling recommending optimal insertion sites based on patient anatomy
- Automated documentation capturing insertion parameters for medical records

Case Study: Selecting a Pen Needles Manufacturer – A Distributor’s Journey
Background: MedSupply Solutions, a regional medical device distributor in Southeast Asia, sought to expand their product portfolio to include safety pen needles. They had three finalist manufacturers:
- Manufacturer A: Low-cost Chinese producer, minimal certifications, MOQ 500,000 units
- Manufacturer B: Mid-tier European manufacturer, CE marked, MOQ 100,000 units, 30% higher pricing
- Manufacturer C: Established Asian manufacturer with FDA and CE approvals, MOQ 200,000 units, competitive pricing
Evaluation Process:
- Sample Testing: Ordered 100 units from each manufacturer for hands-on evaluation
- Manufacturer A: Inconsistent needle sharpness, 8% retraction mechanism failures
- Manufacturer B: Excellent quality, but wing design too rigid for local clinician preferences
- Manufacturer C: Consistent quality, culturally appropriate design based on regional market feedback
- Facility Audit: Conducted virtual factory tours
- Manufacturer A: Declined virtual tour, provided stock photos instead (red flag)
- Manufacturer B: Comprehensive tour showing automated production, but single production line posed capacity concerns
- Manufacturer C: Multiple production lines, robust quality lab, cleanroom environments
- Regulatory Review: Verified certification authenticity
- Manufacturer A: Claimed ISO 13485 but could not provide certificate number for third-party verification
- Manufacturer B: Valid certifications but no presence in target Asian markets
- Manufacturer C: Full regulatory package including ASEAN Medical Device Directive compliance
- Pilot Order: 50,000 units to test real-world performance
- Manufacturer C selected based on balanced risk-reward profile
- Delivered on-time with zero defects reported by hospital customers
- Responsive technical support resolved minor packaging labeling questions
Outcome: MedSupply Solutions established a long-term partnership with Manufacturer C, growing from 50,000 units in year one to 2.5 million units annually within three years. The relationship expanded to include custom-branded products and co-development of a specialized neonatal safety pen needle.
Key Lessons:
- Lowest price doesn’t equal lowest total cost when factoring in quality failures and customer complaints
- Regulatory credentials require verification, not just acceptance of supplier claims
- Cultural fit and market-specific design knowledge matter for regional distribution success

Conclusion: Making an Informed Decision
Selecting the right safety pen needle supplier requires balancing multiple factors: product quality, regulatory compliance, manufacturing capacity, pricing, and long-term partnership potential.
Decision Checklist for Buyers:
✅ Verify Certifications: Confirm ISO 13485, FDA, CE, and other relevant approvals through independent verification
✅ Test Product Quality: Order samples and conduct side-by-side comparisons with competitor products
✅ Assess Manufacturing Capability: Evaluate production capacity, lead times, and scalability to meet your growth trajectory
✅ Understand Total Cost: Calculate complete landed cost including freight, duties, and potential quality failure expenses
✅ Review Contract Terms: Negotiate clear terms for pricing, payment, intellectual property, and dispute resolution
✅ Plan for Growth: Ensure the supplier can accommodate 2-3x volume increases as your distribution expands
✅ Build Relationships: Establish regular communication cadences with account managers and technical support
By applying this comprehensive framework, healthcare distributors, hospital procurement teams, and medical device buyers can confidently select pen needles manufacturers that deliver superior products, reliable supply chains, and lasting value.
About Kohope Medical
As a leading manufacturer of safety-engineered medical devices, Kohope Medical specializes in producing high-quality safety pen needles and comprehensive needle solutions for healthcare providers worldwide. Our ISO 13485 certified facilities combine advanced manufacturing technology with rigorous quality systems to deliver products that meet the highest international standards.
Why Choose Kohope Medical:
- ✓ Full regulatory compliance: FDA registered, CE marked, ISO certified
- ✓ Comprehensive product range: 18G-26G gauges, multiple lengths and configurations
- ✓ Custom OEM/ODM services with flexible MOQ options
- ✓ Proven track record supplying 50+ countries across six continents
- ✓ Dedicated technical support and clinical training programs
Contact us today for product samples, detailed specifications, or to discuss your specific requirements. Our team of medical device specialists is ready to help you find the perfect safety pen needle solution for your clinical applications.