In modern healthcare and home diagnostics, the blood lancet has become an indispensable tool for capillary blood sampling. From routine diabetes monitoring to clinical diagnostics, these small but sophisticated devices play a critical role in patient care. This comprehensive guide explores the technology, safety features, and clinical applications of modern safety lancets, with particular focus on advanced pressure activated lancet systems and ergonomic square shape lancet designs.
What Is a Blood Lancet?
A medical lancet is a single-use, sterile device designed to puncture the skin for capillary blood collection. Unlike traditional needles used for venipuncture, a disposable lancet is specifically engineered for shallow, controlled penetration of the skin’s outer layers to obtain small blood samples.
Primary Functions
- Capillary blood collection for glucose monitoring
- Sample acquisition for diagnostic testing
- Point-of-care blood analysis
- Home healthcare applications
Modern automatic blood lancet devices have evolved significantly from their predecessors, incorporating advanced safety mechanisms and user-friendly designs that minimize pain and maximize protection.

Understanding Safety Lancet Technology
The Evolution of Safety Features
Traditional lancets posed significant risks, including accidental needle-stick injuries and cross-contamination. The development of safety blood lancet technology addressed these concerns through innovative engineering.
Key Safety Mechanisms
Pressure-Activated Systems
A pressure activated lancet operates through contact-activation technology. The needle remains concealed until the device is pressed against the skin with appropriate pressure. This mechanism ensures:
- No premature needle exposure
- Activation only upon intentional use
- Immediate retraction after sampling
Auto-Retractable Technology
The auto retractable lancet feature represents a major advancement in needlestick injury prevention. Upon completion of the puncture, the needle automatically withdraws into the device housing, rendering it permanently safe. This design eliminates the possibility of reuse or accidental contact.
Sterile Packaging and Single-Use Design
Each sterile blood lancet is individually sealed in sterile packaging, maintaining aseptic conditions until the moment of use. The single-use nature of these devices fundamentally prevents cross-contamination between patients.

The Square Shape Lancet: Ergonomic Innovation
Design Rationale
The square lancet represents an ergonomic advancement over traditional cylindrical designs. This geometric configuration offers several practical advantages:
Enhanced Grip and Control
The flat surfaces of a square shape lancet provide tactile reference points, allowing healthcare professionals to maintain precise control during the procedure. This is particularly important when working with pediatric patients or individuals with limited finger space.
Reduced Rolling Risk
Unlike round devices that can roll off surfaces, the square configuration remains stable on flat surfaces, reducing the risk of contamination or loss during preparation.
Improved Positioning Accuracy
The defined edges of the square design enable more consistent perpendicular positioning against the skin, which contributes to:
- More predictable penetration depth
- Reduced tissue trauma
- Improved first-attempt success rates
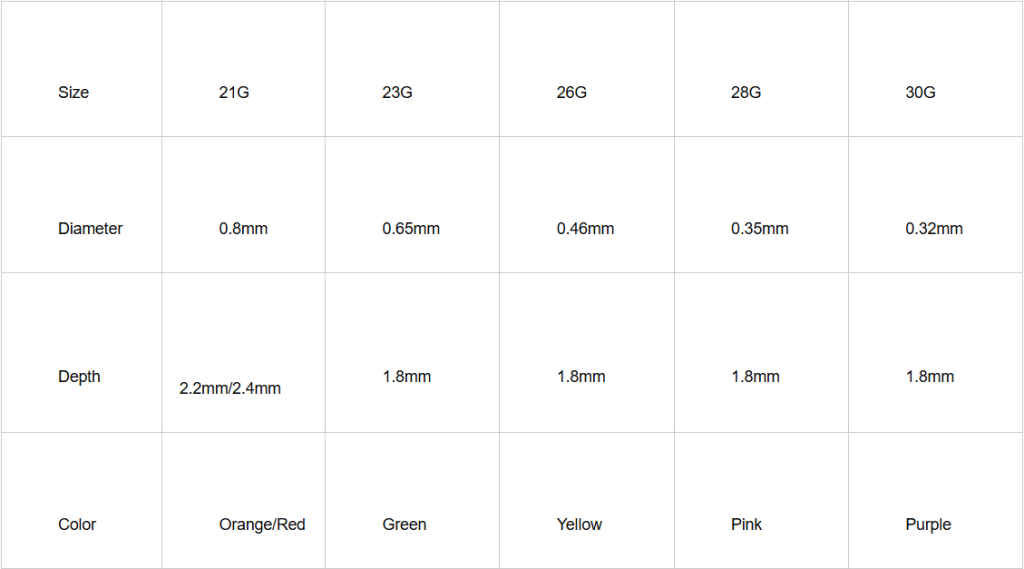
Gauge Selection: 21G vs. 23G Blood Lancets
Understanding needle gauge is essential for selecting the appropriate blood collection lancet for specific applications.
Gauge Measurement System
The gauge system operates inversely: a higher gauge number indicates a thinner needle. Both 21g blood lancet and 23g blood lancet options serve distinct clinical purposes.
21G Blood Lancet
Specifications:
- Larger needle diameter
- Deeper penetration capability
- Greater blood flow volume
Optimal Applications:
- Patients with thicker skin
- Tests requiring larger sample volumes
- Clinical laboratory settings
23G Blood Lancet
Specifications:
- Finer needle diameter
- Shallower penetration depth
- Reduced tissue disruption
Optimal Applications:
- Routine diabetes lancet testing
- Pediatric patients
- Individuals with sensitive skin
- Frequent daily monitoring



Clinical Applications and Use Cases
Diabetes Management
The most widespread application of the modern lancing device is in diabetes blood glucose monitoring. Millions of individuals worldwide rely on these devices for daily, sometimes multiple-times-daily, blood sugar testing.
Requirements for Diabetes Lancets:
- Minimal pain to encourage compliance
- Consistent blood sample volume
- Easy single-handed operation
- Reliable performance across multiple daily uses
Hospital and Clinical Settings
In professional healthcare environments, medical supplies lancet devices must meet stringent standards:
- Diagnostic Laboratories: Standardized blood collection for various tests
- Emergency Departments: Rapid point-of-care glucose assessment
- Pediatric Units: Low-trauma sampling for young patients
- Geriatric Care: Safe operation for elderly patients with fragile skin
Home Healthcare
The simplicity and safety features of modern safety lancets have made home blood monitoring accessible to millions. The one-step operation process—twist, position, press—requires no specialized training while maintaining clinical-grade safety standards.
Proper Usage Protocol
Step-by-Step Procedure
Preparation:
- Wash hands thoroughly with soap and water
- Select an appropriate puncture site (typically the side of a fingertip)
- Remove the sterile blood lancet from its sealed package
Activation:
- Twist and remove the protective cap
- Position the device firmly against the selected site
- Apply gentle, steady pressure until activation occurs
Post-Procedure:
- The auto retractable lancet mechanism engages automatically
- Apply gentle pressure to the puncture site if needed
- Dispose of the used device in an approved sharps container
Safety Considerations
- Never reuse a disposable lancet
- Do not share devices between individuals
- Verify sterile packaging integrity before use
- Follow local regulations for sharps disposal


Selecting a Blood Lancet Supplier
Quality Indicators
When sourcing from a blood lancet supplier, healthcare organizations and distributors should evaluate:
Regulatory Compliance:
- FDA clearance or CE marking
- ISO 13485 certification (medical device quality management)
- Regular compliance audits and documentation
Manufacturing Standards:
- Controlled cleanroom environments
- Validated sterilization processes
- Batch testing and traceability systems
Product Consistency:
- Uniform penetration depth across production lots
- Reliable activation mechanism performance
- Consistent sterility assurance levels
OEM and Custom Solutions
Many institutions require OEM blood lancet services for branded programs or specialized applications. A qualified safety lancet manufacturer should offer:
- Custom packaging and labeling
- Specification modifications for unique requirements
- Regulatory support for market-specific compliance
- Technical consultation for product selection

Comparative Analysis: Lancets vs. Traditional Methods
Advantages of Modern Safety Lancets
Pain Reduction: The ultra-sharp, beveled needles of contemporary blood lancet devices, combined with rapid penetration and immediate retraction, create significantly less discomfort than older methods. This encourages better patient compliance, particularly important for individuals requiring frequent monitoring.
Infection Control: Single-use, sterile blood lancet systems eliminate cross-contamination risks entirely. The sealed sterile packaging maintains aseptic conditions until the moment of use.
Operational Efficiency: The simple activation mechanism of a pressure activated lancet reduces procedure time and training requirements. Healthcare workers can perform blood sampling quickly and safely, even in high-volume settings.
Safety Profile: The auto retractable lancet mechanism provides permanent needlestick protection. Once activated, the device cannot cause accidental injuries, protecting both healthcare workers and patients.
Regulatory Landscape and Standards
International Requirements
Medical lancet devices are classified as medical devices and must comply with regional regulatory frameworks:
United States:
- FDA 510(k) clearance required
- Classification as Class II medical device
- Good Manufacturing Practice (GMP) compliance
European Union:
- CE marking under Medical Device Regulation (MDR)
- Conformity assessment procedures
- Technical documentation requirements
International:
- ISO 13485 quality management certification
- ISO 23908 specific standards for lancets
- Regional import and distribution licensing
Quality Assurance Testing
Reputable manufacturers of safety lancets conduct rigorous testing:
- Sterility validation through microbial challenge studies
- Penetration depth consistency verification
- Activation force standardization
- Shelf-life stability studies
- Packaging integrity testing

Future Developments in Lancet Technology
Emerging Innovations
Smart Integration: Next-generation lancing device systems may incorporate:
- Bluetooth connectivity for automatic data logging
- Integration with continuous glucose monitoring systems
- Usage tracking for compliance monitoring
Advanced Materials: Research into novel needle coatings promises:
- Further pain reduction through enhanced lubrication
- Antimicrobial surfaces for additional safety
- Improved penetration consistency
Customized Depth Control: Variable-depth mechanisms may allow:
- User-adjustable penetration settings
- Automatic depth selection based on skin thickness
- Optimized sampling for different body sites
Conclusion
The modern blood lancet represents a sophisticated integration of safety engineering, ergonomic design, and clinical effectiveness. From the pressure activated lancet mechanism that prevents premature needle exposure to the square shape lancet configuration that enhances control, these devices reflect decades of innovation focused on patient comfort and safety.
Whether for routine diabetes lancet testing at home or professional diagnostic applications in clinical settings, selecting the appropriate device requires understanding the interplay of gauge selection, safety features, and ergonomic considerations. Healthcare providers and organizations should partner with qualified blood lancet supplier companies that demonstrate consistent quality, regulatory compliance, and commitment to innovation.
As technology continues to advance, we can expect even greater refinement in safety blood lancet design, further reducing patient discomfort while maintaining the rigorous safety standards that protect both healthcare workers and patients.
Explore our complete range of safety lancets →
References and Further Reading
For healthcare professionals seeking additional technical information about blood collection lancet devices, consult:
- ISO 23908:2011 – Sharps injury protection requirements
- CLSI GP42-A6 – Procedures for collection of capillary blood specimens
- FDA guidance documents on safety-engineered medical devices
- CDC guidelines for infection control in healthcare settings