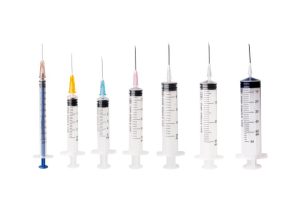
The global cosmetic industry is experiencing a transformative shift with the widespread adoption of glass prefilled syringes. These precision-engineered devices have become the gold standard for delivering dermal fillers, botulinum toxins, and other injectable aesthetic treatments across North America, Europe, Japan, and South Korea. As a specialized manufacturer, Kohope delivers prefilled syringe solutions that meet the stringent requirements of developed markets.

Global Market Trends for Cosmetic Prefilled Syringes
Market Size and Growth Projections
The glass prefilled syringe market for cosmetic applications reached $1.8 billion in 2023 and is projected to grow at a 8.5% CAGR through 2030, reaching $3.2 billion. This growth is driven by increasing demand for minimally invasive aesthetic procedures.
Regional Market Breakdown:
- North America: 40% market share, 250+ million units annually
- Europe: $650 million market value, strict EU MDR compliance requirements
- Asia-Pacific: Fastest-growing region at 12% annual growth, led by South Korea and Japan
Key Consumer Trends
Developed markets show three critical preferences:
- Ready-to-Use Convenience – Pre-sterilized prefilled syringes reduce clinic preparation time by 60%
- Precision Dosing – Accuracy within ±0.01ml for consistent treatment outcomes
- Enhanced Safety – Anti-counterfeiting features and full traceability systems
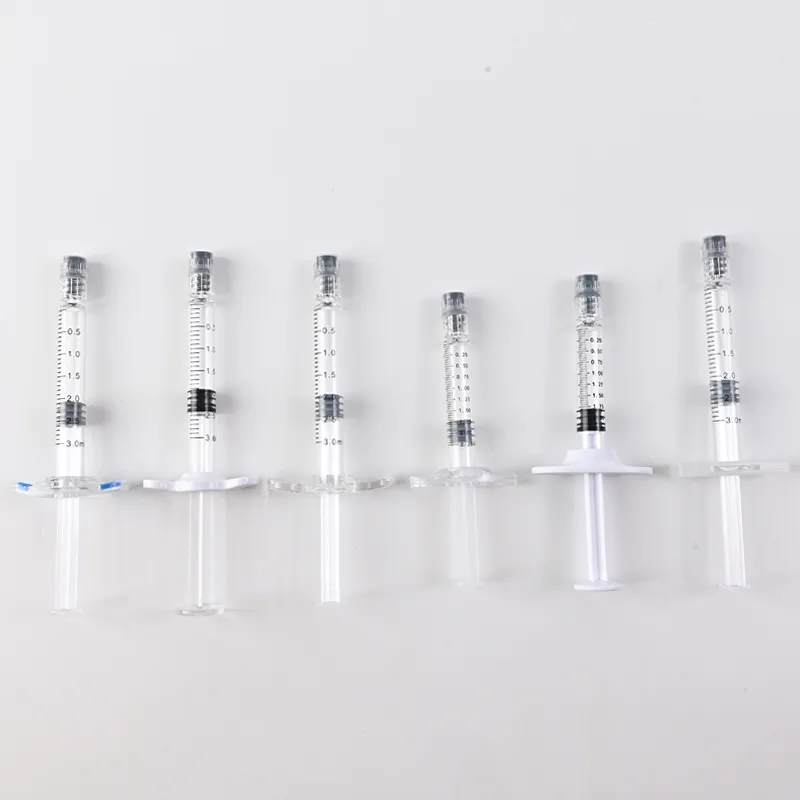
Technical Advantages of Glass Prefilled Syringes
Superior Material Performance
Type I Borosilicate Glass Benefits:
- Chemical Inertness: Alkali leaching <50ppb ensures compatibility with peptides and growth factors
- Optical Clarity: >90% transparency for visual inspection during injection
- Barrier Properties: Zero oxygen permeability protects oxygen-sensitive cosmetic ingredients
Advanced Siliconization:
Kohope’s proprietary surface treatment reduces friction coefficient to <0.15, ensuring smooth plunger operation with injection force controlled at 15-25N for optimal physician control.
Precision Manufacturing Standards
Volume Specifications for Cosmetic Applications:
- 1ml – Fine detail work (lips, tear troughs)
- 2-3ml – Mid-volume treatments (nasolabial folds, cheeks)
- 5ml – Large-area contouring (body sculpting)
Manufacturing Tolerances:
- Internal diameter: ±0.02mm
- Graduation accuracy: ±2%
- Residual volume: <0.1ml (67% less than conventional syringes)
Sterility Assurance
ISO 13485-Certified Production:
Kohope operates Class 100,000 cleanrooms with ETO sterilization achieving SAL 10⁻⁶ (Sterility Assurance Level), exceeding FDA and EU requirements.
Seal Integrity:
Medical-grade bromobutyl rubber stoppers ensure 5-year shelf life with zero leakage or contamination.

Kohope’s Manufacturing Excellence
Production Capabilities
Scale and Efficiency:
- Annual capacity: 50+ million units
- Automation rate: 85%
- Lead time: 30% faster than industry average
Global Certifications:
✓ ISO 13485 Medical Device Quality Management
✓ FDA DMF (Drug Master File) registration
✓ CE Mark under EU MDR 2017/745
✓ Korea MFDS approval
✓ Japan PMDA certification
Market-Specific Solutions
For North American Markets:
- USP-compliant borosilicate glass specifications
- Complete E&L (Extractables & Leachables) testing data
- DMF cross-referencing support for accelerated product approvals
For European Markets:
- Full compliance with EU MDR technical documentation requirements
- Batch-level traceability systems
- Multi-language labeling capabilities
For Asian Markets (Korea & Japan):
- Flexible production for 0.5ml-3ml popular formats
- Rapid development cycles (3-6 months)
- Defect rate: <50ppm (parts per million)
Innovation and Sustainability
Smart Integration Features:
- NFC Chip Technology for product lifecycle tracking
- Anti-counterfeit Labels using color-shifting inks
- Ergonomic Design for enhanced grip stability
Environmental Commitment:
- 100% recyclable glass components
- Biodegradable packaging materials
- Carbon Neutrality Goal: 50% emission reduction by 2030
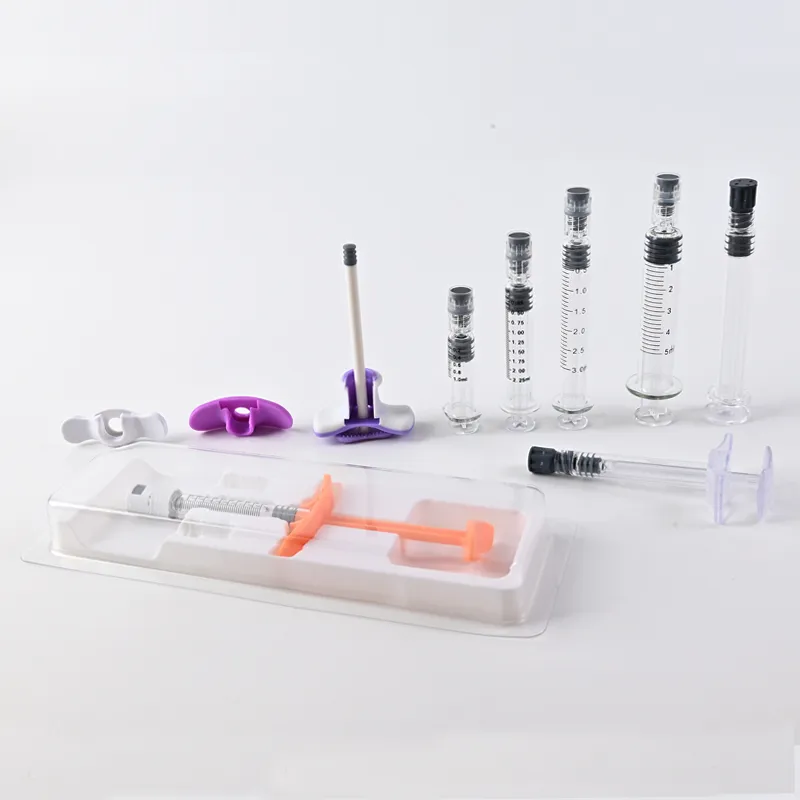
Clinical Applications and Performance Data
Hyaluronic Acid Filler Market
The global HA filler market reached $5.8 billion in 2023, with 80% utilizing prefilled syringes. Kohope supplies major international brands with documented performance:
- Product complaint rate: <10ppm
- Physician satisfaction: 95%+
- Injection precision improvement: 40% better than traditional assembled syringes
Botulinum Toxin Applications
Korea and the US lead global botulinum toxin consumption, demanding strict quality controls:
- Silicone oil residue: <0.5mg per unit
- Particulate matter (≥10μm): 0 particles per unit
- Approved by TOP 5 global neurotoxin brands


Industry Outlook and Recommendations
Future Technology Directions
Next-Generation Innovations:
- Advanced Biocompatible Coatings – Reduced protein adsorption
- Smart Injection Systems – Pressure sensors for depth and speed feedback
- Sustainable Alternatives – Eco-friendly materials balancing performance and environmental impact
Guidance for Cosmetic Brands
Supplier Selection Criteria:
✓ Verify comprehensive regulatory certifications for target markets
✓ Assess production capacity and delivery reliability
✓ Evaluate customization capabilities for specific product formulations
Quality Partnership Indicators:
- Long-term supply chain stability
- Technical support for regulatory submissions
- Continuous improvement programs
Conclusion
As the global aesthetic medicine industry advances toward standardization, precision, and intelligence, glass prefilled syringes serve as critical medical devices ensuring safety and efficacy. Kohope combines 20+ years of manufacturing expertise with rigorous quality control and continuous innovation to deliver world-class prefilled syringe for cosmetic applications to North America, Europe, Japan, and Korea.
Our commitment: Safer treatments, higher efficiency, sustainable development.
About Kohope
Professional Glass Prefilled Syringe Manufacturer
- 20+ years industry experience
- Serving 50+ countries worldwide
- Specialized solutions for cosmetic and pharmaceutical industries
📧 Contact our business team for detailed technical documentation and certification materials.
Data sourced from published market research and industry statistics. All specifications subject to customization based on client requirements.