The disposable sterile hypodermic syringe has revolutionized modern healthcare, evolving from a reusable luxury to an indispensable single-use medical device. As healthcare facilities worldwide prioritize patient safety and infection control, understanding the development, market dynamics, and optimal specifications of medical syringes has become crucial for procurement professionals and healthcare administrators.
The Evolution of Hypodermic Syringes: From Innovation to Essential Medical Device
Early Development (1850s-1950s)
The hypodermic syringe’s journey began in the mid-19th century when Irish physician Francis Rynd developed the first hollow needle for subcutaneous medication delivery in 1844. By 1853, Alexander Wood in Scotland and Charles Pravaz in France independently refined the design, creating the prototype of modern syringes. These early instruments were crafted from glass and metal, requiring extensive sterilization and reuse—a practice that unknowingly contributed to cross-contamination and disease transmission.
For nearly a century, reusable glass syringes dominated medical practice. Healthcare workers meticulously boiled and autoclaved these instruments between patients, a time-consuming process that couldn’t eliminate all infection risks. The hepatitis B and HIV/AIDS epidemics of the 20th century dramatically highlighted the dangers of inadequate sterilization.
The Disposable Revolution (1950s-1980s)
The introduction of the disposable sterile hypodermic syringe in the 1950s marked a watershed moment in medical safety. New Zealand pharmacist Colin Murdoch invented the first practical disposable syringe in 1956, combining plastic manufacturing technology with sterile packaging innovations. His design addressed critical safety concerns while dramatically reducing healthcare costs associated with sterilization labor and equipment.
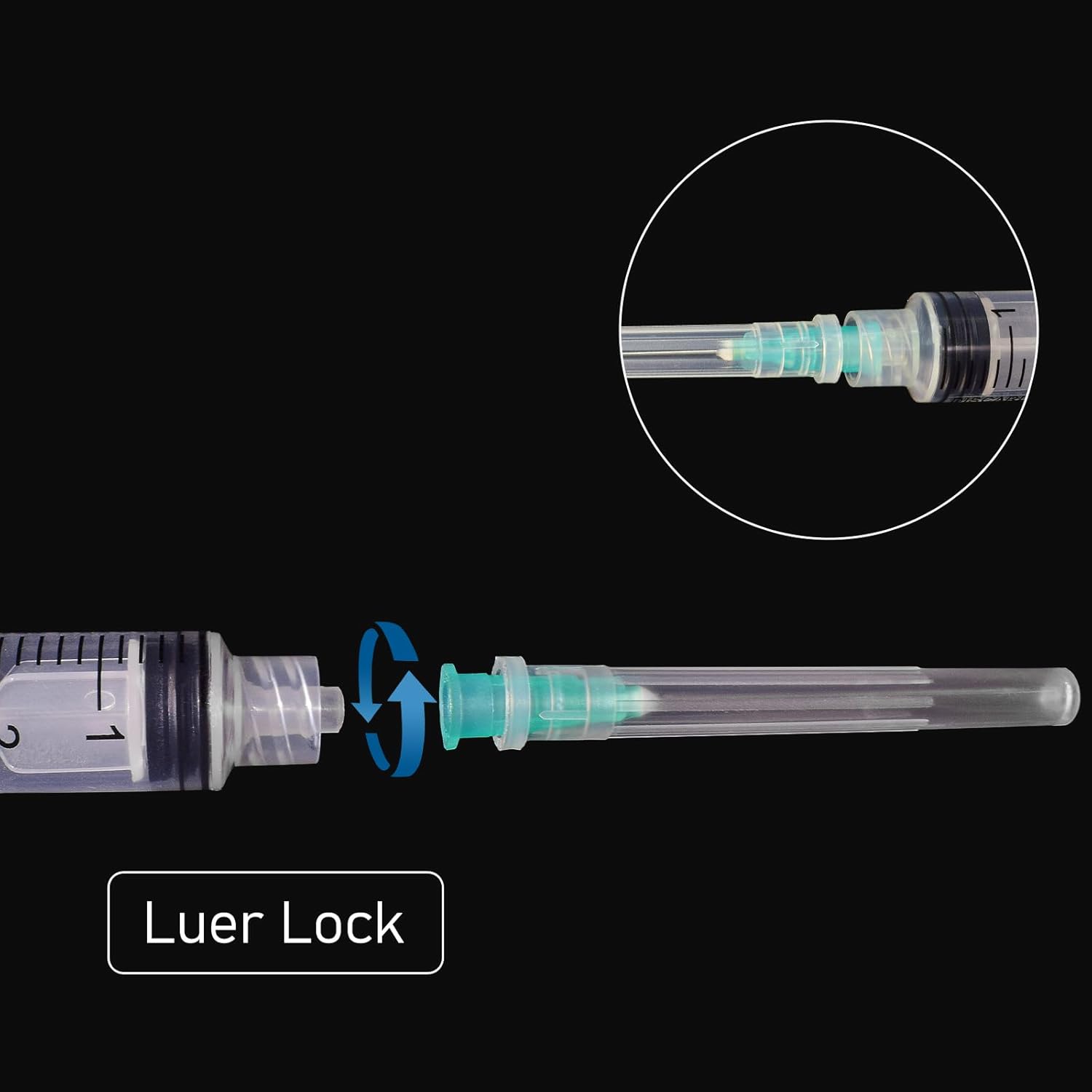
By the 1960s, Becton Dickinson and other manufacturers began mass-producing plastic disposable sterile hypodermic syringes, making them economically viable for widespread adoption. The 1970s saw the introduction of the Luer Lock connection system, which provided secure needle attachment—a feature now standard in products like the 5ml disposable sterile hypodermic syringe with 21G needle.
Modern Innovations (1990s-Present)
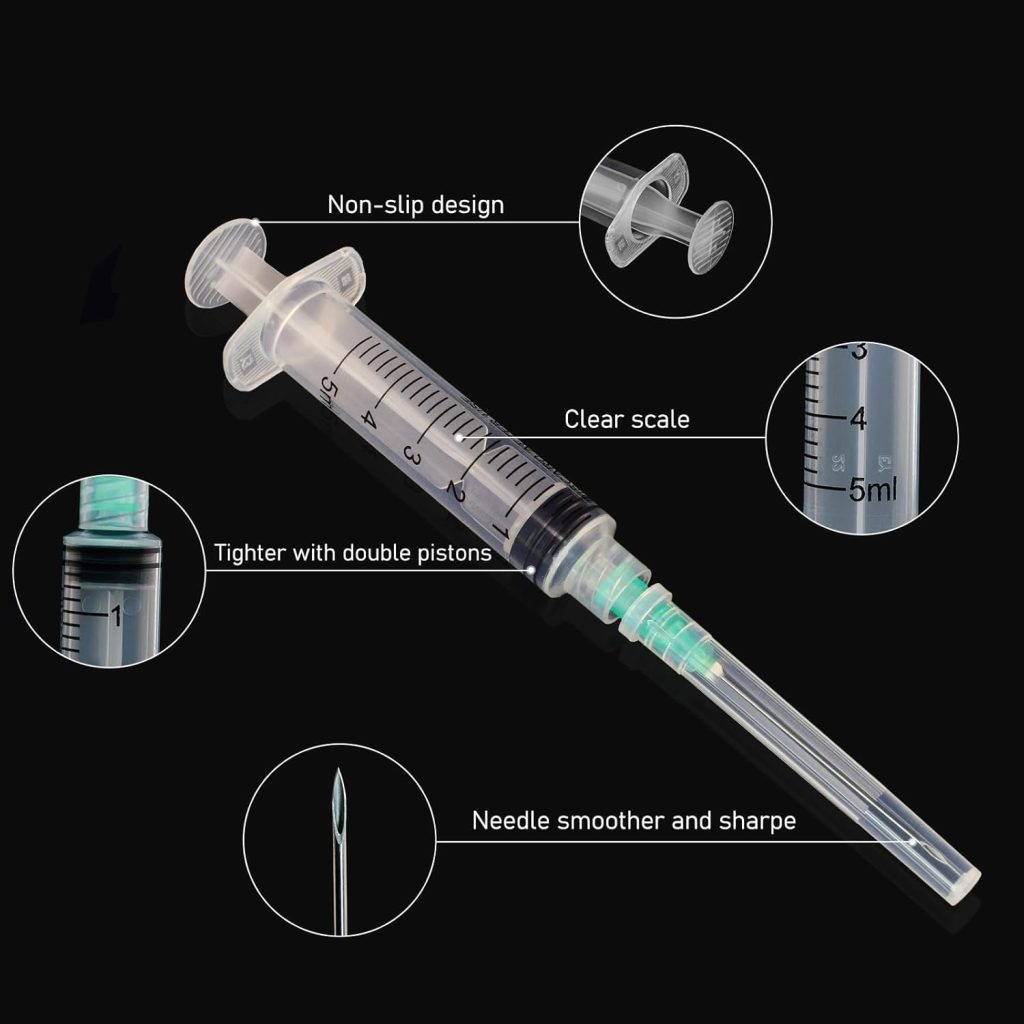
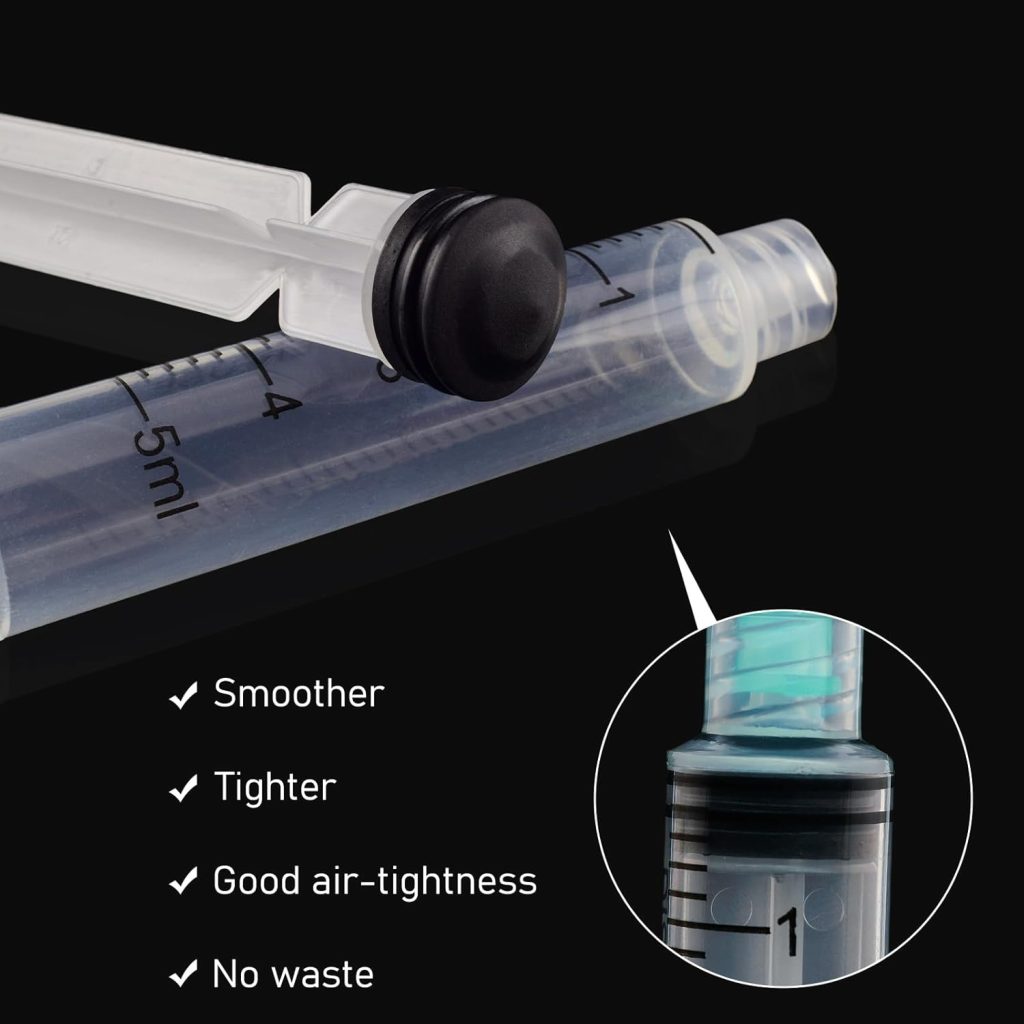
The late 20th and early 21st centuries brought significant refinements: safety-engineered devices with retractable needles, auto-disable mechanisms for vaccination campaigns, and precision manufacturing that ensures consistent quality. Today’s disposable sterile hypodermic syringes feature graduated scales accurate to 0.1ml, latex-free materials for allergy prevention, and individually wrapped sterile packaging that guarantees five-year shelf stability.
Global Market Size and Growth Trajectory
Current Market Valuation
The global disposable syringes market has experienced exponential growth, reflecting increased healthcare access and safety awareness worldwide. As of 2024, the market is valued at approximately $18.2 billion USD, with projections indicating it will reach $28.5 billion by 2031, representing a compound annual growth rate (CAGR) of 6.8%.
The disposable sterile hypodermic syringe segment accounts for roughly 65% of this market, with conventional syringes (non-safety engineered) representing the largest volume due to their cost-effectiveness and versatility across medical applications.
Regional Market Distribution
North America leads global consumption with approximately 32% market share, driven by stringent infection control regulations, advanced healthcare infrastructure, and high per-capita medical expenditure. The United States alone consumes over 7 billion syringes annually across hospitals, clinics, and outpatient facilities.
Europe accounts for 28% of the global market, with Germany, France, and the UK representing the largest consumers. EU Medical Device Regulation (MDR) compliance has elevated quality standards, benefiting manufacturers who maintain ISO 13485 certification.
Asia-Pacific represents the fastest-growing regional market with 9.2% CAGR, driven by expanding healthcare infrastructure in China, India, and Southeast Asian nations. Government immunization programs and increasing chronic disease prevalence fuel demand for disposable sterile hypodermic syringes across diverse capacity ranges.
Market Segmentation by Syringe Capacity and Specifications
Understanding market distribution across different syringe sizes helps procurement professionals optimize inventory and budget allocation.
1ml Syringes: Precision Medication and Insulin Delivery
Market Share: 22% of total volume
The 1ml syringe segment serves specialized applications requiring precise, small-volume medication delivery. Insulin-dependent diabetics represent the largest consumer group, with over 537 million diabetic patients worldwide requiring daily injections. Additionally, 1ml syringes with 25G-27G needles dominate pediatric vaccination programs and tuberculin skin testing.
Key Applications:
- Insulin administration for diabetes management
- Subcutaneous hormone injections (growth hormone, fertility treatments)
- Intradermal allergy testing
- Pediatric medication dosing
- Tuberculosis screening (Mantoux test)
Market Value: Approximately $4.0 billion annually
3ml Syringes: Versatile Clinical Workhorse
Market Share: 35% of total volume
Three-milliliter syringes represent the most widely used capacity across general medical practice. Their versatility makes them ideal for routine intramuscular and subcutaneous injections, vaccine administration, and blood sample collection when paired with appropriate needles.
Key Applications:
- Routine vaccinations (influenza, COVID-19, hepatitis)
- Intramuscular antibiotic administration
- Vitamin B12 and other supplement injections
- Venipuncture and blood collection
- General medication delivery in outpatient settings
Market Value: Approximately $6.4 billion annually
The 3ml segment’s dominance reflects its optimal balance between medication capacity and patient comfort, particularly for vaccines requiring 0.5-1.0ml doses where the extra barrel space facilitates easier aspiration and injection technique.
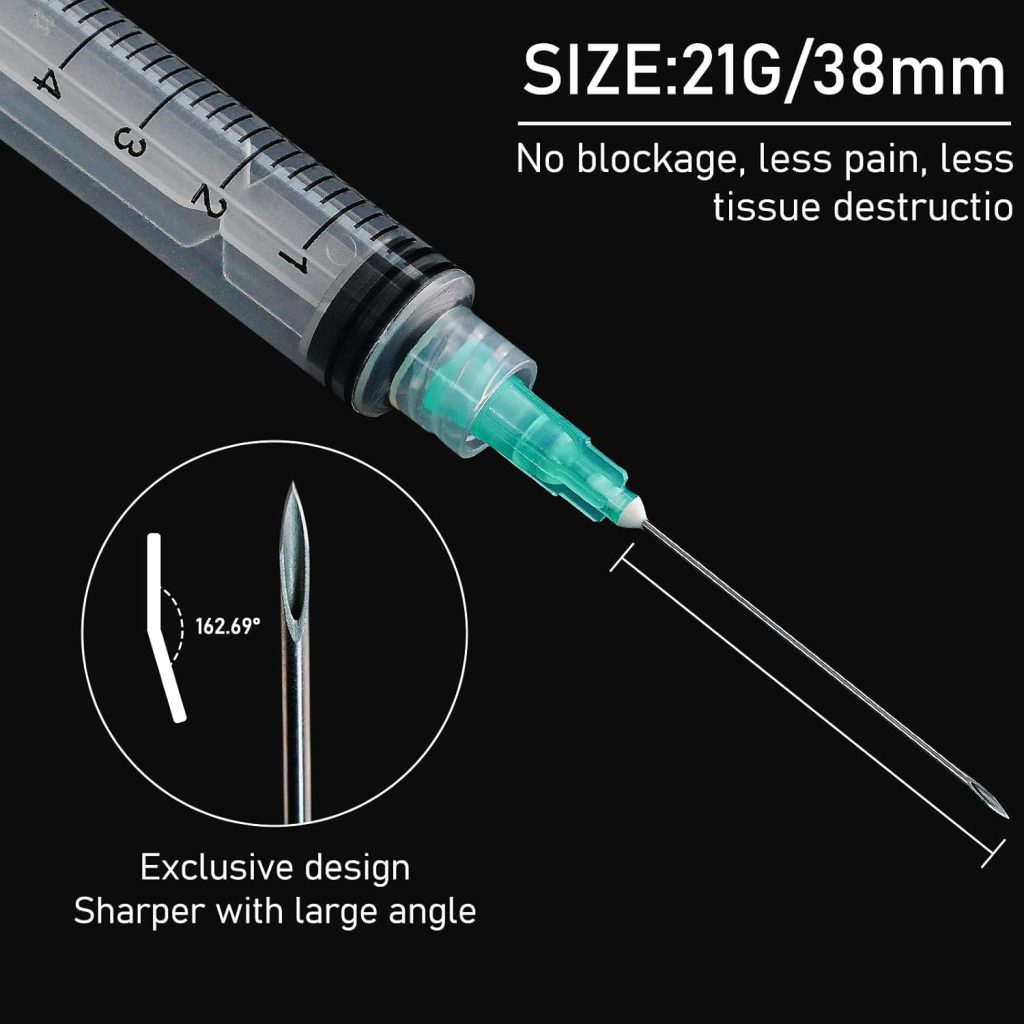
5ml Syringes: Mid-Range Medical Applications
Market Share: 18% of total volume
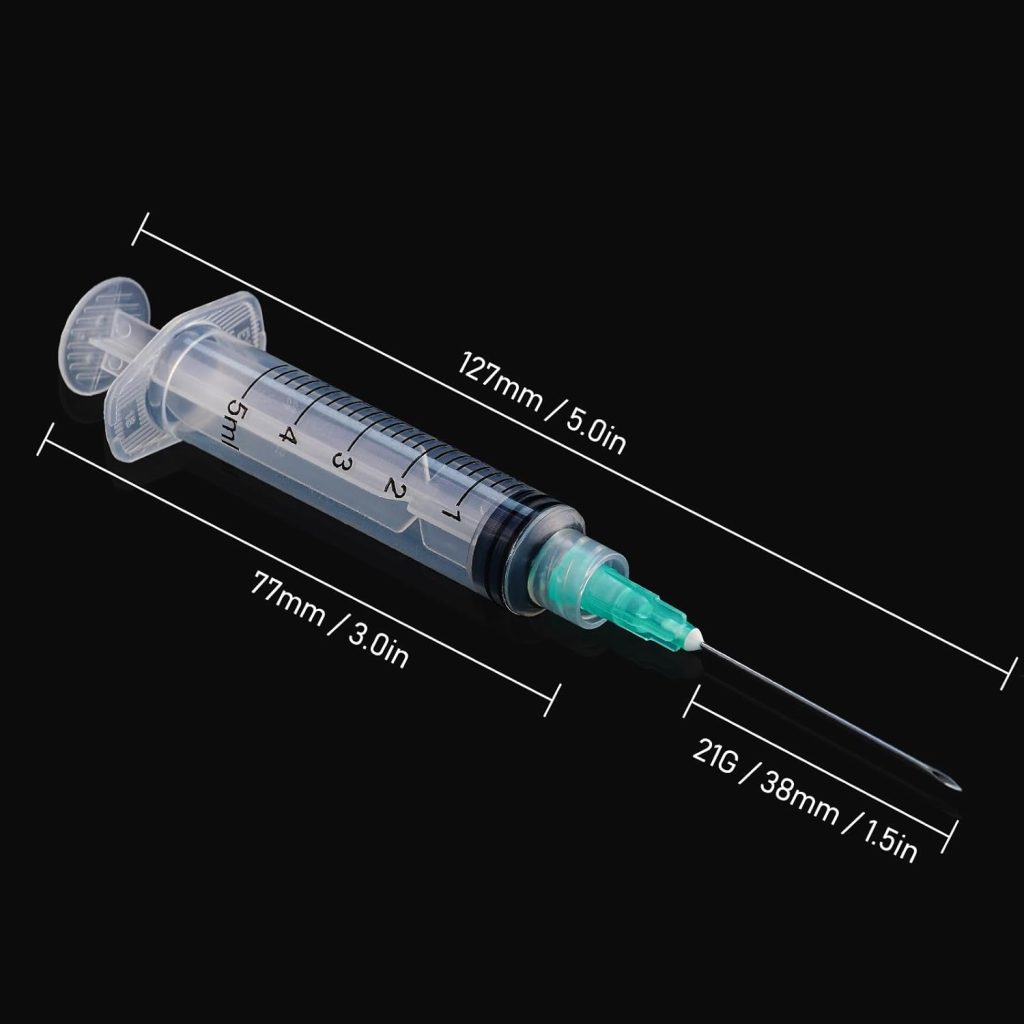
The 5ml disposable sterile hypodermic syringe occupies a critical mid-range position, ideal for applications requiring larger medication volumes or viscous solutions. When equipped with 21-gauge needles, these syringes excel at intramuscular injections in adult patients and therapeutic procedures requiring deeper tissue penetration.
Key Applications:
- Intramuscular injections in gluteal or vastus lateralis sites
- Administration of viscous medications (certain antibiotics, oil-based preparations)
- Therapeutic joint injections (corticosteroids)
- Blood draw procedures
- Emergency medication delivery (naloxone, epinephrine in larger doses)
- Contrast media injection for imaging procedures
Market Value: Approximately $3.3 billion annually
Technical Considerations:
The 5ml disposable sterile hypodermic syringe with 21G 1.5-inch needle represents an optimal configuration for adult intramuscular injections. The 21-gauge (0.8mm) diameter provides sufficient bore for viscous medications while minimizing patient discomfort, and the 1.5-inch (38mm) length ensures proper medication delivery into muscle tissue for patients with normal body composition.
10ml and Larger Syringes: High-Volume Applications
Market Share: 15% of total volume
Larger capacity disposable sterile hypodermic syringes (10ml, 20ml, 50ml, and 60ml) serve specialized applications requiring substantial fluid volumes. These syringes typically feature Luer Lock connections to ensure secure attachment during high-pressure injections or fluid aspiration.
Key Applications:
- Irrigation procedures (wound cleaning, catheter flushing)
- Enteral feeding administration
- Contrast media injection for CT and MRI imaging
- Large-volume medication reconstitution
- Drainage of fluid collections
- Laboratory sample preparation
Market Value: Approximately $2.7 billion annually
Safety-Engineered Syringes: Growing Premium Segment
Market Share: 10% of total volume (rapidly growing)
Safety syringes with retractable needles or protective shields represent the premium segment, commanding 40-60% price premiums over conventional disposable sterile hypodermic syringes. Regulatory mandates in developed markets and WHO recommendations for immunization programs drive adoption, particularly in North America and Europe where needlestick injury prevention legislation requires safety-engineered devices.
Market Value: Approximately $1.8 billion annually, with projected 11% CAGR through 2030
Needle Gauge Selection: Matching Specifications to Clinical Needs
Proper needle gauge selection significantly impacts patient comfort, medication delivery efficacy, and procedural success rates. Understanding gauge-specific applications optimizes clinical outcomes.
Fine Gauge Needles (25G-30G): Minimal Tissue Trauma
Applications: Subcutaneous injections, insulin delivery, intradermal procedures, pediatric vaccinations
These ultra-fine needles minimize pain and tissue trauma, making them ideal for frequent injections (diabetes management) and sensitive patient populations (children, elderly with fragile skin). However, their narrow bore limits use to low-viscosity medications.
Medium Gauge Needles (21G-23G): Versatile Clinical Applications
Applications: Intramuscular injections, blood collection, most vaccinations
The 21-gauge needle featured in our 5ml syringe represents the clinical sweet spot for adult IM injections. Its 0.8mm diameter accommodates moderately viscous medications while maintaining reasonable patient comfort. The 1.5-inch length ensures proper deltoid or gluteal muscle penetration in most adult patients.
23-gauge needles (0.6mm) serve similar purposes with slightly reduced flow rates but improved patient comfort, often preferred for elderly patients or those with lower pain tolerance.
Large Gauge Needles (18G-20G): Viscous Medications and Rapid Delivery
Applications: Viscous antibiotic administration, rapid fluid replacement, blood donation, emergency medications
Larger bore needles facilitate faster flow rates essential for emergency situations and accommodate highly viscous solutions (certain penicillin formulations, oil-based preparations). However, increased tissue trauma necessitates careful technique and appropriate patient selection.

Manufacturing Excellence and Quality Standards
Modern disposable sterile hypodermic syringe production represents a pinnacle of precision manufacturing, combining automated assembly with rigorous quality control.
Production Process
Contemporary syringe manufacturing utilizes injection molding technology to create barrels and plungers from medical-grade polypropylene. ISO Class 7 or Class 8 cleanroom environments prevent particulate contamination during assembly. Automated needle attachment systems ensure consistent Luer Lock connection torque, while inline vision systems verify graduated scale accuracy and identify defects.
Sterilization typically employs ethylene oxide (ETO) gas, achieving Sterility Assurance Level (SAL) of 10⁻⁶—meaning less than one in one million syringes could theoretically harbor viable microorganisms. Individual blister packaging under controlled humidity conditions protects sterility and extends shelf life to five years.
Regulatory Compliance
Quality disposable sterile hypodermic syringes from reputable manufacturers maintain multiple certifications:
- ISO 13485:2016 Medical Device Quality Management System certification
- FDA 21 CFR Part 820 compliance for US market access
- EU MDR 2017/745 conformity for European distribution
- CE marking demonstrating safety and performance standards
- WHO Prequalification for international procurement eligibility
Manufacturers implement comprehensive traceability systems, enabling complete batch tracking from raw material receipt through distribution—critical for recall management and adverse event investigation.
Current Market Trends Shaping Procurement Decisions
Sustainability Initiatives
Environmental concerns increasingly influence procurement decisions. Manufacturers now explore biodegradable polymers and recyclable packaging alternatives. Some healthcare systems implement syringe waste segregation programs to reduce environmental impact, though single-use devices remain essential for infection control.
Supply Chain Resilience
The COVID-19 pandemic exposed vulnerabilities in global medical supply chains, prompting healthcare organizations to diversify supplier bases and maintain larger safety stock. Procurement professionals now prioritize manufacturers with proven production capacity and geographic diversification.
Digital Integration
Smart syringes with RFID tracking and integrated dose monitoring represent emerging technologies, particularly valuable for expensive biologic medications and clinical trial applications. While currently niche, these innovations may reshape inventory management and medication administration documentation.
Combination Products
Pre-filled disposable sterile hypodermic syringes containing ready-to-administer medications reduce medication errors and preparation time. This segment grows at 8.5% CAGR, particularly for emergency medications (epinephrine, naloxone) and high-value biologics.

Procurement Best Practices for Healthcare Organizations
Volume Forecasting and Inventory Optimization
Accurate consumption forecasting prevents both stockouts and excess inventory carrying costs. Analyze historical usage patterns by department, considering:
- Seasonal vaccination campaigns (influenza, back-to-school immunizations)
- Chronic disease patient populations requiring regular injections
- Emergency department patient volume trends
- Surgical procedure schedules
- Anticipated regulatory changes affecting safety device requirements
Implement par level systems with automated reorder points, maintaining 30-60 days safety stock for critical items like the 5ml disposable sterile hypodermic syringe while minimizing slow-moving inventory.
Supplier Evaluation Criteria
When selecting disposable sterile hypodermic syringe suppliers, assess:
Quality Assurance:
- ISO 13485 certification and regulatory compliance documentation
- Batch testing protocols and certificates of analysis availability
- Defect rates and customer complaint resolution processes
- Sterility validation and stability testing data
Supply Reliability:
- Manufacturing capacity and backup production facilities
- Lead time consistency and on-time delivery performance
- Inventory holding practices and emergency fulfillment capabilities
- Geographic diversification reducing geopolitical risk
Commercial Terms:
- Volume-based pricing tiers and annual contract discounts
- Payment terms and early payment incentive programs
- Consignment inventory or vendor-managed inventory options
- Price protection clauses during contract term
Technical Support:
- Product training and clinical education resources
- Adverse event reporting and investigation protocols
- Regulatory documentation support for Joint Commission compliance
- Custom packaging or private labeling capabilities
Cost-Effectiveness Analysis
While unit cost drives many procurement decisions, total cost of ownership provides more accurate comparison:
Direct Costs:
- Unit purchase price
- Shipping and handling fees
- Inventory carrying costs (storage space, obsolescence risk)
Indirect Costs:
- Clinical staff time for product evaluation and training
- Quality control and incoming inspection labor
- Waste disposal fees (particularly for safety-engineered devices)
- Potential costs of product failures (patient injury, litigation, reputation damage)
Premium products like individually wrapped disposable sterile hypodermic syringes may command 5-10% price premiums but deliver value through reduced contamination risk, extended shelf life, and improved inventory management flexibility.

Standardization vs. Variety
Balancing standardization benefits against clinical need diversity requires careful analysis. Excessive SKU proliferation increases inventory complexity, while insufficient variety forces clinicians to compromise on optimal specifications.
Recommended Approach:
- Standardize on 3-5 core syringe configurations covering 85-90% of clinical needs
- Maintain the 5ml disposable sterile hypodermic syringe with 21G needle as standard for adult IM injections
- Stock 1ml and 3ml syringes for subcutaneous and pediatric applications
- Keep specialized sizes (10ml+, safety-engineered) for specific departmental needs
- Establish formulary exception processes for unusual clinical requirements
Group Purchasing Organization (GPO) Considerations
GPO contracts typically deliver 10-25% savings versus direct purchasing for smaller healthcare facilities. However, evaluate:
- Contract compliance requirements and administrative fees
- Volume commitment obligations
- Product substitution flexibility during shortages
- Local vs. contract pricing for high-volume items
- Ability to negotiate directly for facility-specific needs
Large health systems may achieve better pricing through direct manufacturer negotiation, particularly for high-volume items like disposable sterile hypodermic syringes.

Emerging Markets and Future Opportunities
Asia-Pacific Growth Drivers
Expanding middle-class populations in India, China, Indonesia, and Vietnam drive healthcare infrastructure investment. Government immunization programs and chronic disease management initiatives create sustained demand for disposable sterile hypodermic syringes. Manufacturers establishing regional production facilities reduce logistics costs and tariff exposure while improving delivery reliability.
African Healthcare Expansion
The African continent represents significant long-term growth potential as healthcare access improves. International donor programs (GAVI, WHO) and domestic health initiatives drive vaccination coverage expansion, creating demand for reliable, cost-effective disposable sterile hypodermic syringes. Auto-disable syringes gain traction in immunization programs to prevent reuse.
Personalized Medicine Impact
Biologic medications and personalized therapies increasingly require specialized delivery devices. While pre-filled syringes dominate biologic delivery, reconstitution and administration still rely on conventional disposable sterile hypodermic syringes, particularly the 3ml and 5ml capacities suited to typical biologic dose volumes.
Final Procurement Recommendations
Healthcare procurement professionals seeking optimal disposable sterile hypodermic syringe solutions should consider the following strategic approach:
1. Establish Core Product Standards
Designate the 5ml disposable sterile hypodermic syringe with 21G 1.5-inch needle and Luer Lock as your standard for adult intramuscular medication administration. This specification delivers versatility across therapeutic applications while maintaining cost-effectiveness and clinical efficacy.
2. Prioritize Quality Over Price
The marginal cost difference between premium and economy disposable sterile hypodermic syringes pales compared to potential costs of product failures, patient complications, or regulatory non-compliance. Specify manufacturers with:
- ISO 13485:2016 certification demonstrating quality management system maturity
- FDA registration and compliance with 21 CFR Part 820
- Complete lot traceability and certificates of analysis for every shipment
- Proven track record in healthcare institutional supply
3. Implement Risk-Based Inventory Strategy
Maintain 45-60 days safety stock for critical items like the 5ml syringe used across multiple departments. Consider dual-sourcing strategies for mission-critical specifications, balancing cost efficiency against supply continuity assurance.
4. Leverage Data for Continuous Improvement
Track clinical outcomes, staff satisfaction, and cost metrics across different disposable sterile hypodermic syringe specifications. Engagement with frontline clinical staff ensures product selections align with workflow realities and patient care quality objectives.
5. Plan for Regulatory Evolution
Anticipate increasing requirements for safety-engineered devices, sustainability documentation, and supply chain transparency. Partner with manufacturers demonstrating innovation commitment and regulatory compliance investment.

Conclusion
The disposable sterile hypodermic syringe represents one of modern medicine’s most significant safety advances, transforming infection control and enabling safe medication delivery across billions of patient encounters annually. From its 19th-century origins to today’s precision-manufactured devices, the hypodermic syringe evolution reflects healthcare’s broader commitment to patient safety and clinical excellence.
Understanding market dynamics, specification requirements, and procurement best practices empowers healthcare organizations to optimize their syringe supply strategies. Whether selecting the versatile 5ml disposable sterile hypodermic syringe with 21G needle for general clinical use or specialized configurations for specific applications, informed decision-making drives both clinical quality and economic efficiency.
As healthcare continues evolving toward personalized medicine, digital integration, and enhanced safety, the fundamental role of quality disposable sterile hypodermic syringes remains constant—delivering medications safely, reliably, and cost-effectively to patients worldwide.