Quick Navigation:
Key Differences: Animal vs. Human AV Fistula Needles
Can They Be Shared? Professional Analysis
Primary Application Environments
Kohope’s Professional Recommendations
Manufacturing Standards: The Kohope Advantage
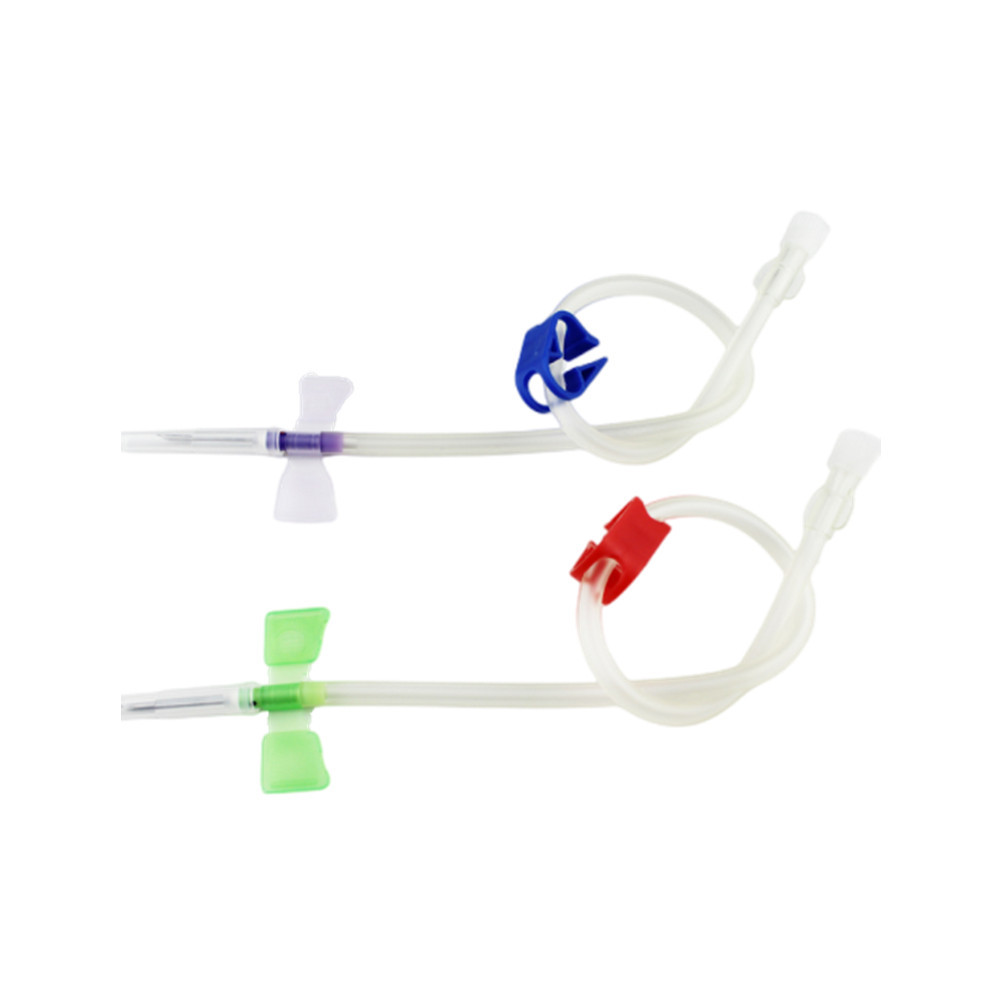
As a leading AV fistula needle manufacturer, Kohope understands the critical distinctions between animal and human medical devices. AV fistula needles are specialized medical instruments designed for hemodialysis vascular access. While both veterinary and human healthcare utilize these devices, the question of cross-application requires professional clarification.
Key Differences: Animal vs. Human AV Fistula Needles
Design Specifications
Human AV Fistula Needles
- Standardized gauge sizes (typically 15-17G)
- Precision-engineered for human vascular anatomy
- Consistent length specifications for adult patients
Animal AV Fistula Needles
- Variable sizing for different species (small cats to large livestock)
- Adapted needle angles for diverse vascular structures
- Customized configurations based on animal physiology
Regulatory Standards & Biocompatibility
At Kohope, we manufacture AV fistula needles under distinct regulatory frameworks:
Human Medical Devices
- FDA/CE certification mandatory
- Sterility Assurance Level (SAL) of 10⁻⁶
- Rigorous human biocompatibility testing (ISO 10993)
Veterinary Medical Devices
- Veterinary-specific regulatory compliance
- Species-appropriate material validation
- Flexible standards while maintaining safety
Can They Be Shared? Professional Analysis
❌ Animal-Grade Needles for Human Use: Absolutely Prohibited
Kohope emphasizes that veterinary AV fistula needles must never be used on humans. Here’s why:
Regulatory Violations
- Breaches medical device legislation globally
- Lacks human-grade certification
Safety Risks
- Potentially inadequate sterilization standards
- Unverified human biocompatibility
- Infection and adverse reaction risks
Ethical Concerns
- Violates medical ethics principles
- Compromises patient safety standards
✓ Human-Grade Needles for Animal Use: Conditionally Acceptable
In specific scenarios, human-grade AV fistula needles may be utilized in veterinary settings:
When Appropriate:
- Emergency veterinary interventions
- High-end pet dialysis treatments
- Appropriate size match for the animal’s vasculature
Advantages:
- Superior sterility standards exceed veterinary requirements
- Enhanced safety margin for animal patients
- Proven biocompatibility from human medical standards
Primary Application Environments
Human Healthcare Facilities
Dialysis Centers Kohope’s AV fistula needles serve chronic kidney disease patients requiring:
- Regular hemodialysis (2-3 sessions weekly)
- Arteriovenous fistula access
- Consistent, reliable vascular puncture
Hospital Nephrology Departments
- Acute kidney injury management
- Emergency blood purification procedures
- Toxicology treatment protocols
Veterinary & Research Settings
Veterinary Hospitals Modern pet healthcare increasingly utilizes dialysis needles for:
- Feline and canine renal failure treatment
- Companion animal hemodialysis programs
- Advanced veterinary nephrology services
Research Laboratories AV fistula needles support scientific research in:
- Pharmacokinetic studies
- Toxicology experiments
- Biomedical vascular access modeling
- Large animal research protocols
Wildlife Rescue Centers
- Emergency blood purification for poisoned wildlife
- Critical care interventions
- Conservation medicine applications
Kohope’s Professional Recommendations
For Human Healthcare Providers
✓ Exclusively use certified human medical-grade AV fistula needles ✓ Select manufacturers with comprehensive traceability systems ✓ Verify FDA/CE certification before procurement ✓ Maintain strict single-use protocols
For Veterinary Professionals
✓ Primary choice: Veterinary-specific dialysis needles ✓ Alternative option: Human-grade devices (when specifications match) ✓ Ensure appropriate gauge selection for target species ✓ Never repurpose veterinary devices for potential human contact
For Research Institutions
✓ Match AV fistula needle specifications to research protocols ✓ Comply with Institutional Animal Care and Use Committee (IACUC) requirements ✓ Document device usage and disposal procedures ✓ Source from reputable manufacturers with research-grade certifications
Manufacturing Standards: The Kohope Advantage
As a specialized AV fistula needle manufacturer, Kohope maintains:
Quality Assurance
- ISO 13485 certified manufacturing
- Dual production lines (human/veterinary grade)
- Rigorous quality control protocols
- Complete batch traceability
Product Innovation
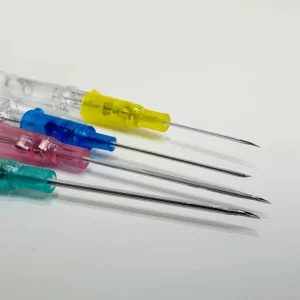
- Tri-bevel needle tip technology for minimal trauma
- Ultra-thin wall design maximizing flow rates
- Ergonomic grip for precise puncture control
- Color-coded gauge identification
Safety Features
- Individual sterile packaging
- Clear usage indicators
- Latex-free materials
- Back-eye for smooth insertion
Conclusion
AV fistula needles designed for animals and humans cannot be interchangeably used due to fundamental regulatory, safety, and ethical differences. While human-grade dialysis needles may serve veterinary applications under specific conditions, the reverse is categorically prohibited.
Kohope, as a professional AV fistula needle manufacturer, advocates for:
- Appropriate device selection based on end-user requirements
- Regulatory compliance in all application scenarios
- Never compromising safety for cost considerations
- Species-specific device utilization as best practice
Whether for human dialysis centers, veterinary hospitals, or research laboratories, selecting the correct AV fistula needle ensures optimal outcomes, regulatory compliance, and patient safety.
Contact Kohope for professional consultation on AV fistula needle selection tailored to your specific clinical or research requirements. Our technical team provides expert guidance ensuring you choose the right device for your application.