As a professional medical device manufacturer, Kohope understands that many customers and end-users have questions about the differences between amber oral syringes and standard medical syringes. This article provides a detailed analysis from multiple perspectives—including use cases, design philosophy, manufacturing processes, and material selection—to help you understand the essential distinctions between these two products.

I. Core Difference: Fundamental Design Purpose
Amber Oral Syringe
Design Purpose: Specifically engineered for accurate measurement and oral administration of liquid medications, particularly for light-sensitive formulations.
Key Features:
- Needle-free design preventing injection misuse
- ENFit/ISO 80369-3 connector incompatible with injection systems
- Amber-colored material protecting photosensitive medications
- Capacity range: typically 1ml-20ml for pediatric and precision dosing
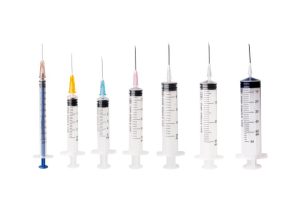
Standard Medical Syringe
Design Purpose: Designed for parenteral administration via subcutaneous, intramuscular, or intravenous injection.
Key Features:
- Luer connector compatible with various needles
- Transparent material for visualizing medication and air bubbles
- Capacity range: 1ml-60ml
- Sterile packaging with strict requirements
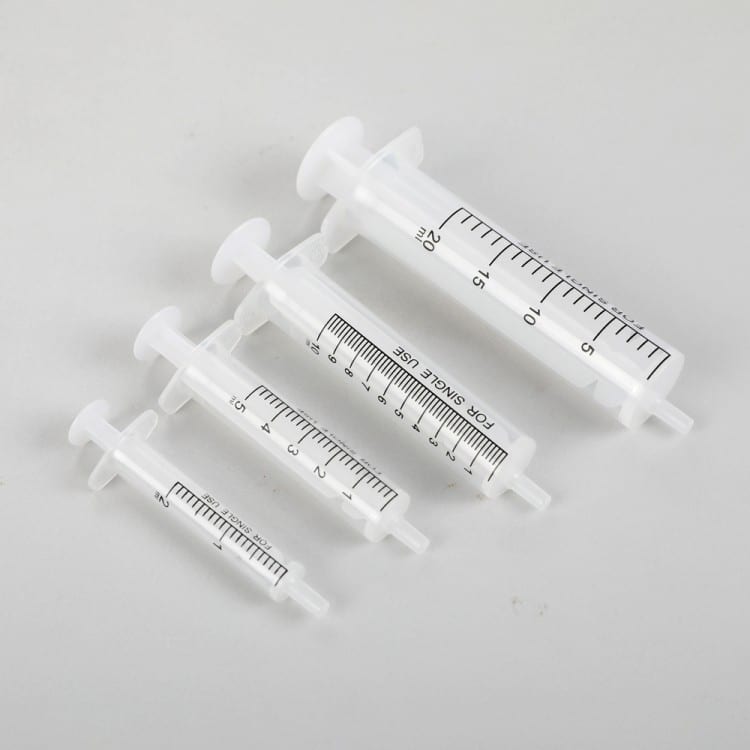
II. Application Scenarios and Requirements
Amber Oral Syringe Applications
Pediatric Medication The amber oral syringe excels in delivering precise 0.5ml-5ml liquid doses to children, eliminating the 20-30% dosage error associated with household spoons while preventing accidental needle ingestion hazards.
Photosensitive Drug Administration The amber syringe provides critical protection for light-sensitive medications including vitamin A and D preparations, certain antibiotic suspensions, and oral chemotherapy formulations. The amber material blocks over 90% of visible and UV light.
Home Healthcare Perfect for elderly patients requiring precise oral medication, patients with dysphagia receiving tube feeding, and veterinary applications where the amber oral design ensures safety and accuracy.
Clinical Nutrition Support Healthcare professionals use amber oral syringes for precise feeding through nasogastric tubes and gastrostomy tubes, enabling accurate control of feeding rate and dosage.

Standard Medical Syringe Applications
Standard syringes serve clinical injection therapy (IV, IM, SC, ID routes), vaccine administration, blood collection and laboratory testing, and emergency medication delivery requiring rapid intravenous access and sterile operation.
III. Design and Manufacturing Differences
Safety Design Philosophy
Amber Oral Syringe Design Core principle: Never Events prevention through physical isolation—the connector is completely incompatible with injection systems. International standards mandate amber coloring with purple plunger, “ORAL USE ONLY” labeling, and cone or duckbill-shaped tips that cannot connect to needles.
Manufacturing Standards:
- ISO 20698: Single-use oral syringe standard
- ISO 80369-3: Small-bore connector standard (ENFit)
- FDA 21 CFR 880.5860: Oral dosing device requirements
Standard Medical Syringe Design Core principle: Sterility and precision with Luer taper connector (ISO 594), sterile assurance level (SAL) 10^-6, and ±5% graduation accuracy per ISO 7886-1.
Manufacturing Standards:
- ISO 7886-1: Single-use sterile syringe standard
- ISO 594: Luer taper connector standard
- FDA 21 CFR 880.5860: Syringe device requirements
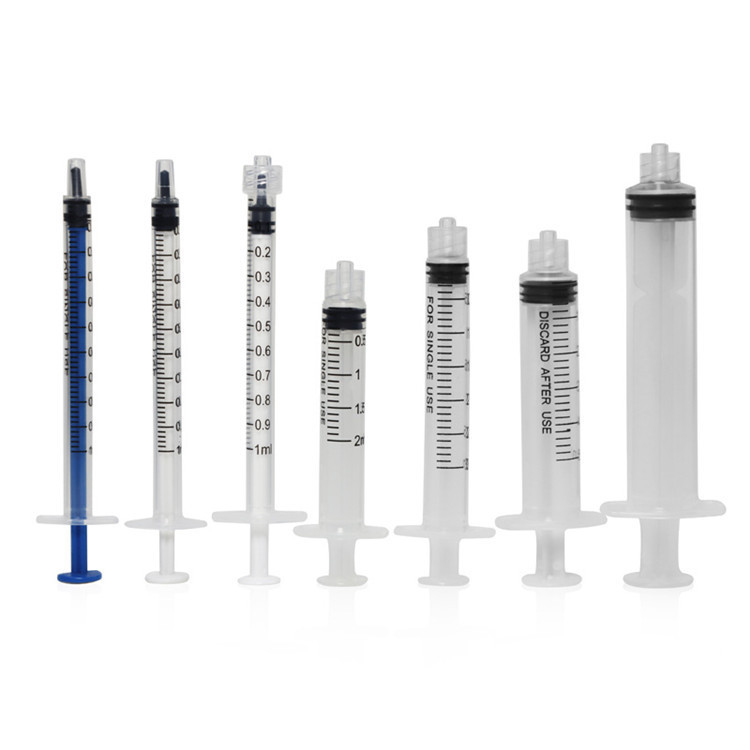
Material Selection Science
Amber Oral Syringe Material System
| Component | Material | Properties | Selection Rationale |
|---|---|---|---|
| Barrel | Amber PP | >90% light blocking | Protects photosensitive drugs |
| Plunger | Purple PE or TPE | Friction coefficient 0.15-0.25 | Color differentiation + smooth action |
| Seal | Medical-grade silicone/TPE | USP Class VI biocompatibility | Ensures sealing, non-toxic |
| Tip | PP | FDA food-grade certified | Oral contact safety |
Critical Technical Parameters for Amber Syringes:
- Light transmission: <10% at 400-700nm wavelength
- Extractables testing: Meets USP <661> plastics requirements
- Biocompatibility: ISO 10993 series compliance
- Chemical resistance: pH 2-12, alcohol-resistant
Standard Medical Syringe Material System
| Component | Material | Properties | Selection Rationale |
|---|---|---|---|
| Barrel | Clear PP or medical PS | >92% transparency | Visual inspection of medication |
| Plunger | Natural rubber or TPE | Superior sliding performance | Precise injection control |
| Hub | PP | High strength | Secure needle connection |
| Seal | Natural/synthetic rubber | Excellent sealing | Prevents leakage |
Critical Technical Parameters:
- Sterile assurance level: SAL 10^-6
- Endotoxin level: <0.5 EU/ml
- Graduation accuracy: ±5% (ISO 7886-1)
- Sliding force: <20N (10ml specification)
- Residual volume: <0.1ml (low dead space syringes)
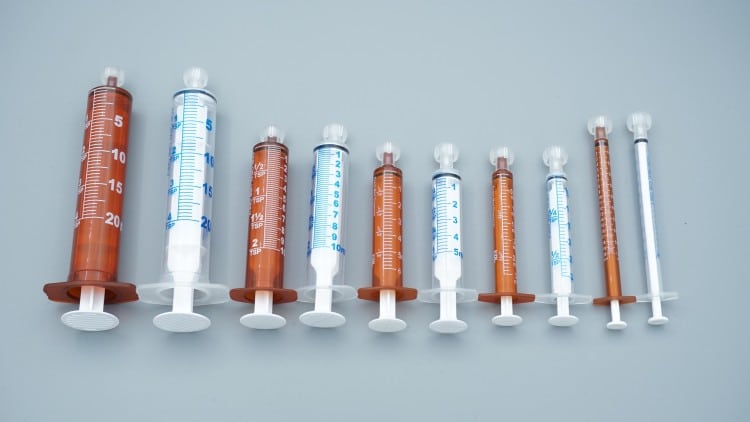
Manufacturing Process Differentiation
Kohope’s Amber Oral Syringe Production
Our amber oral production line processes raw materials with 2-5% amber masterbatch addition, drying PP at 80°C for 4 hours with batch spectral analysis for light-blocking performance. Injection molding operates at 220-260°C with ±0.02mm mold precision and ENFit connector tolerance of ±0.05mm. Assembly occurs in Class 100,000 cleanrooms (non-sterile) with automated plunger insertion and cap sealing, maintaining push force <15N.
Quality control for the amber syringe includes ±3% graduation accuracy (exceeding ISO standards), spectrophotometer light-blocking tests, positive/negative pressure leak testing, and 1.2-meter drop tests.
Standard Medical Syringe Production
Production utilizes medical-grade materials meeting USP Class VI or ISO 10993 standards in climate-controlled storage. Injection molding occurs in Class 10,000 cleanrooms with laser graduations precise to ±0.01mm and Luer connectors meeting ISO 594 (6% taper). Sterilization uses ethylene oxide (EO) or gamma irradiation (25-50 kGy) with biological indicator validation.
Quality control encompasses sterility testing (USP <71>), endotoxin testing (LAL method), mechanical sliding performance, and residual volume verification <0.1ml.
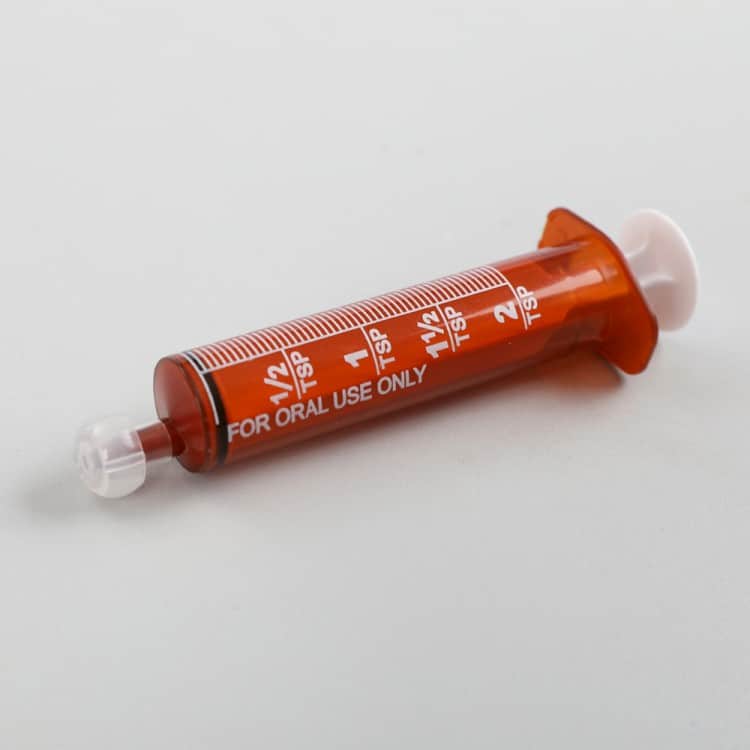
IV. Regulatory and Certification Requirements
Amber Oral Syringe Certifications
- CE Marking: MDR 2017/745 (Class I device)
- FDA Registration: 510(k) exempt category
- ISO 20698: Oral syringe-specific standard
- ISO 80369-3: ENFit connector certification
Testing includes ISO 10993-5 (cytotoxicity), 10993-10 (sensitization), USP <661> chemical testing, and functional testing (graduation accuracy, push force, sealing) without sterility requirements unless specifically labeled.
Standard Medical Syringe Certifications
- CE Marking: MDR 2017/745 (Class IIa with needle or Class I sterile)
- FDA: 510(k) clearance
- ISO 7886-1: Syringe standard
- ISO 13485: Quality management system
Testing requires full ISO 10993 biocompatibility battery, USP <71> sterility, USP <85> endotoxin testing, and functional validation of sliding performance, Luer connector strength, and graduation accuracy.
V. Cost Structure and Market Analysis
Cost Comparison (5ml Example)
Amber Oral Syringe
- Raw materials: $0.08-0.12 (amber PP 15-20% premium over clear)
- Tooling: $15,000-25,000 (complex ENFit connector molds)
- Production: $0.05-0.08
- Packaging: $0.02-0.03 (non-sterile packaging)
- Unit factory price: $0.15-0.25
Standard Medical Syringe
- Raw materials: $0.06-0.10
- Tooling: $12,000-20,000
- Production: $0.05-0.08
- Sterilization: $0.03-0.05 (EO sterilization)
- Packaging: $0.03-0.05 (sterile packaging)
- Unit factory price: $0.18-0.30
Market Trends
The amber oral syringe market reaches approximately $450-500 million globally (2024) with 8-10% CAGR, driven by pediatric medication safety awareness, ENFit standard global adoption (mandatory implementation following 2015 ISO publication), and home healthcare growth. North America leads with 45% market share (ENFit mandatory enforcement), followed by Europe (35%) and Asia-Pacific (15%, rapidly growing).
Standard medical syringes comprise a $15-18 billion global market (2024) growing at 5-6% CAGR, driven by vaccination demands, chronic disease management, and aging populations.
VI. Professional Recommendations from Kohope
For Healthcare Facilities
- Segregated storage: Keep the amber oral syringe separate from injection devices
- Color management: Utilize amber and purple for visual identification
- Standardized training: Ensure staff understands Never Events prevention
- Procurement standards: Prioritize ISO 80369-3 compliant ENFit products
For Pharmaceutical Companies
For photosensitive formulations, we recommend the amber oral syringe as a bundled dosing device. Include specific recommendations for dedicated oral dosing devices in package inserts, and consider incorporating the amber syringe as part of pharmaceutical packaging.
For Home Users
Never use needle-bearing syringes for oral medications—always choose the amber oral syringe for precise dosing, replacing spoons. For photosensitive drugs, select amber syringes for storage and administration. Educate children to recognize “non-injectable” oral syringes.
VII. Innovation and Future Development
Kohope’s Innovation Practice
Our dual-color graduation technology (black primary, red secondary graduations) with magnifying lens raised design improves readability by 30%. Ergonomic improvements include finger rest designs reducing hand fatigue and anti-slip texturing for enhanced grip stability. Environmental material research focuses on bio-based PP reducing carbon footprint by 25% and recyclable single-material designs.
Industry Development Trends
Standardization accelerates with ENFit connector global adoption targeting 80% penetration by 2025 and continuous ISO 80369 series refinement. Smart technology integration includes electronic dose recording, NFC tag traceability, and intelligent reminder systems. Sustainability initiatives pursue 30% plastic reduction, biodegradable material applications, and carbon-neutral production lines.

Conclusion
While the amber oral syringe and standard medical syringe appear similar, they differ fundamentally in design philosophy, application scenarios, manufacturing processes, and material selection. The amber oral focuses on oral administration safety and precision, while standard syringes prioritize injection therapy sterility and reliability.
At Kohope, we maintain our principle of “professional manufacturing, safety first,” continuously innovating technology and improving product quality to contribute to global medical safety. We believe that only through deep understanding of product differences can we ensure correct selection and use, ultimately safeguarding patient safety.
Reference Data Sources:
- ISO 20698:2018 Standard Documentation
- ISO 7886-1:2018 Standard Documentation
- FDA Medical Device Database
- Market Research Reports 2024
- Kohope Internal Quality Control Data
About Kohope: We are a professional medical device manufacturer specializing in oral dosing systems and syringe device R&D and production, with ISO 13485 certification and comprehensive quality management systems. For technical information or product inquiries about our amber oral syringe and other products, please contact our technical team.