In today’s veterinary disposable needles market, precision, safety, and efficiency in animal healthcare medical devices form the cornerstone of quality veterinary medicine. Veterinary disposable needles, while seemingly basic instruments, play a critical role in treatment outcomes, animal welfare, and medical safety standards. These sterile veterinary needles serve as essential tools for veterinary professionals, livestock operators, and procurement decision-makers who require reliable veterinary injection needles for optimal patient care. This comprehensive guide examines every aspect of these essential CE certified veterinary supplies, providing detailed information needed to make informed choices for large animal veterinary equipment and companion animal applications.
1. Product Foundation & Technical Specifications
Product Identity and Core Functionality Analysis
Product Definition: Veterinary Disposable Sterile Injection Needles
The very name of our product encapsulates three fundamental characteristics that define its purpose and value proposition. “Veterinary” signifies specialized design for animal anatomy and physiology; “Disposable” ensures elimination of cross-contamination risks through single-use protocols; and “Sterile” guarantees microbiological safety through validated sterilization processes.
Beyond basic nomenclature, this product represents a sophisticated convergence of materials science, precision manufacturing, and veterinary medical knowledge. Rather than viewing it as a simple animal healthcare medical device, it’s more accurately described as an engineered solution that addresses the complex challenges of animal healthcare delivery.
The functional scope extends across multiple domains. Primary drug delivery capability ensures accurate, complete medication transfer to target tissues within animal patients. The design accommodates various medication viscosities, from low-viscosity aqueous vaccines to high-viscosity oil-based formulations. Secondary diagnostic support functionality facilitates blood collection procedures, requiring careful balance between penetration ease and blood flow characteristics. Tertiary animal welfare protection represents our commitment to minimizing pain and stress through optimized needle geometry and surface treatments.
Professional Positioning Across Three Critical Dimensions:
- Medical Safety Excellence: As devices that directly contact animal tissues and bloodstreams, these veterinary injection needles must exceed the highest medical safety standards. This includes biocompatibility of materials, sterility assurance levels, and infection control capabilities that protect both animal patients and healthcare providers.
- Operational Efficiency Optimization: Veterinary work presents unique challenges including diverse animal physiology, varying cooperation levels, and demanding time pressures. Our design philosophy prioritizes rapid, accurate deployment across all operational conditions while maintaining consistent performance standards.
- Economic Value Delivery: Quality healthcare requires cost-effective solutions. Our products deliver superior cost-benefit ratios by eliminating reprocessing costs, reducing infection risks, and improving operational efficiency—creating measurable value for veterinary practices and agricultural operations.
Target Demographics and Application Scenarios
Large Animal Veterinarians constitute our primary professional user base, serving agricultural and equine sectors. These practitioners manage cattle, horses, sheep, and other livestock in environments ranging from modern veterinary hospitals to field conditions on working farms. They require robust, reliable large animal veterinary equipment capable of consistent performance under demanding conditions. Their preference for 14G and 16G gauges reflects the practical needs of working with large animals that have thick skin and substantial muscle mass.
This user group values durability and penetration power above all else. Field conditions often involve less-than-ideal working environments—dusty barns, outdoor corrals, and time-pressured situations where animals may be uncooperative. The needle must perform flawlessly regardless of these challenges, providing reliable penetration through tough hide and accurate drug delivery to target tissues.
Small Animal Veterinarians represent the fastest-growing segment of our user base, primarily serving companion animal practices. These professionals work in controlled clinical environments with pet owners who are increasingly concerned about their animals’ comfort and welfare. They favor 20G, 22G, and 25G gauges that minimize tissue trauma while maintaining injection accuracy.
This demographic demonstrates sophisticated technical knowledge and high quality expectations. They evaluate sterile veterinary needles based on animal comfort, injection precision, and consistency of performance. Their clientele—pet owners—often requests the highest standard of care regardless of cost, creating demand for premium animal healthcare medical devices.
Agricultural Technicians and Farm Managers handle large-scale vaccination programs and routine healthcare for commercial livestock operations. They require efficient, cost-effective solutions for managing hundreds or thousands of animals. Volume purchasing and standardized protocols characterize their approach, with emphasis on reducing per-animal treatment costs while maintaining safety standards.
Research Institution Personnel demand the highest levels of precision and consistency for experimental protocols. Their applications often require exact dosing, minimal tissue trauma, and reproducible results across multiple subjects. While representing a smaller market segment, this group drives innovation through demanding specifications and detailed feedback.
Detailed Application Environment Analysis:
Clinical Treatment Settings encompass the full spectrum of veterinary medical facilities, from rural mixed practices to specialized urban animal hospitals. These environments require versatile veterinary disposable needles capable of handling diverse cases—from routine wellness care to emergency interventions. Equipment selection must accommodate varying skill levels among staff while maintaining consistent safety standards.
The clinical setting demands rapid instrument changeover, reliable performance under time pressure, and compatibility with existing equipment systems. Practitioners need confidence that their veterinary injection needles will perform consistently whether treating a fractious cat or a cooperative golden retriever.
Preventive Medicine Programs represent high-volume, standardized applications primarily in agricultural settings. Mass vaccination campaigns, herd health protocols, and routine preventive treatments require instruments that maintain performance across hundreds of individual procedures while controlling per-unit costs.
These programs emphasize efficiency and reliability over individual animal comfort, though animal welfare remains important. Standardization of procedures and equipment reduces training requirements and improves consistency across multiple operators.
Blood Collection and Diagnostic Procedures present unique technical challenges requiring specialized performance characteristics. Unlike therapeutic injections, blood collection demands optimized internal diameter for smooth blood flow while minimizing hemolysis and sample contamination.
Diagnostic applications often require multiple samples from individual animals, placing premium value on needle sharpness retention and tissue trauma minimization. Sample quality directly impacts diagnostic accuracy, making needle performance a critical factor in healthcare outcomes.
Technical Specifications and Engineering Excellence
14G Specification (2.108mm OD, 0.152mm Wall Thickness) – Heavy-Duty Performance:
This gauge represents the pinnacle of veterinary disposable needles engineering for large animal veterinary equipment applications. The substantial 2.108mm outer diameter provides exceptional penetration power for thick-skinned animals like cattle and horses, while the robust 0.152mm wall thickness ensures structural integrity under high-force applications.
The internal diameter calculation optimizes flow characteristics for high-viscosity medications commonly used in large animal medicine. Oil-based vaccines, thick antibiotic suspensions, and vitamin preparations flow smoothly through the generously sized internal bore. Length options from 38-76mm accommodate various injection depths, from subcutaneous vaccinations to deep intramuscular treatments in heavily muscled animals.
Manufacturing this gauge requires specialized equipment capable of maintaining dimensional accuracy while working with substantial material thickness. The needle tip geometry receives particular attention—the angle and bevel length are optimized for large animal skin penetration while minimizing the force required for insertion.
16G Specification (1.651mm OD, 0.122mm Wall Thickness) – Versatile Intermediate Solution:
The 16G gauge occupies the critical middle ground in our product line, offering optimal balance between penetration capability and injection comfort for medium-large animals. This specification sees widespread use in swine operations, sheep flocks, and large dog practices.
The engineering challenge with 16G lies in optimizing the wall thickness to internal diameter ratio. Too thick, and internal flow becomes restricted; too thin, and structural integrity suffers. Our 0.122mm wall thickness represents the optimal balance point, validated through extensive flow testing and mechanical stress analysis.
Surface treatment receives enhanced attention at this gauge level. The internal bore undergoes specialized polishing to achieve mirror-like smoothness, critical for maintaining laminar flow of various medication types. External surface treatment includes our proprietary siliconization process, reducing tissue drag during insertion.
18G Through 25G Specifications – Precision Engineering for Diverse Applications:
The progression from 18G to 25G represents increasingly sophisticated manufacturing challenges as dimensions become more precise and tolerance requirements tighten. Each step down in gauge size requires enhanced quality control systems and more advanced production equipment.
At the 25G level (0.514mm OD), we’re approaching the limits of conventional needle manufacturing technology. The 0.064mm wall thickness must be maintained with extreme precision—variations of just a few micrometers can significantly impact performance. This gauge serves the most delicate applications: avian medicine, reptile healthcare, and pediatric animal treatments where tissue trauma must be minimized.
2. Value Proposition and Competitive Advantage
Comprehensive Problem Resolution Analysis
Elimination of Cross-Contamination Risks Through Advanced Design
Healthcare-associated infections represent one of the most significant challenges in veterinary medicine, with potential consequences extending far beyond individual animal welfare. Traditional reusable injection equipment, despite rigorous cleaning and sterilization protocols, carries inherent contamination risks that can compromise entire animal populations.
Our single-use sterile veterinary needles eliminate these risks through fundamental design principles. Each needle undergoes gamma irradiation sterilization to achieve a Sterility Assurance Level (SAL) of 10⁻⁶—meaning less than one unit in a million carries any risk of microbial contamination. This sterilization process penetrates completely through the device, ensuring sterility throughout the entire needle structure.
The packaging system maintains this sterility through multiple barrier protection. Primary packaging creates an immediate sterile barrier, while secondary packaging provides physical protection during storage and transport. The packaging design incorporates tamper-evident features, allowing users to verify that sterility has been maintained from manufacture to use.
Validation of our sterilization process follows ISO 11137 standards, involving biological indicators, chemical indicators, and physical parameter monitoring. We use Geobacillus stearothermophilus spores—among the most radiation-resistant microorganisms—as biological indicators. Only when complete spore kill is demonstrated can we be confident in the sterilization efficacy.
Precision Injection Technology Through Engineering Innovation
Accurate drug delivery requires sophisticated understanding of fluid dynamics, animal anatomy, and injection mechanics. Our multi-gauge veterinary injection needles product line addresses the complex relationships between needle geometry, medication characteristics, and animal physiology.
Internal diameter calculations incorporate advanced fluid dynamics principles, including the Hagen-Poiseuille equation for viscous flow. Each gauge undergoes computational fluid dynamics (CFD) analysis to optimize flow characteristics for medications ranging from low-viscosity saline solutions to high-viscosity oil-based vaccines.
Wall thickness optimization balances multiple competing requirements: structural strength for penetration, internal volume for flow capacity, and material economy for cost effectiveness. Our proprietary wall thickness calculations ensure optimal performance across all gauges while maintaining manufacturing feasibility.
Length specifications address the geometric requirements of various injection techniques. Subcutaneous injections require shorter needles to avoid accidental intramuscular penetration, while deep intramuscular injections need sufficient length to reach target muscle groups. Our length options provide veterinarians with precise tools for each application.
Operational Safety Enhancement Through Systematic Design
Needlestick injuries represent the most common occupational hazard in veterinary medicine, with potential for serious consequences including bloodborne pathogen transmission. Our safety-focused design incorporates multiple protective features to minimize injury risks.
Needle cap design follows ergonomic principles to enable safe single-handed operation. The cap removal force is carefully calibrated—sufficient to prevent accidental removal during handling, yet allowing easy removal when needed. The cap design includes features to prevent reuse, reducing temptation to recap used needles.
Hub design enhances grip security and reduces slippage risk during procedures. The hub dimensions are optimized for human hand proportions, providing secure grip even when hands are wet or gloved. Surface texturing on the hub provides additional grip security without compromising cleanability.
Connection integrity represents a critical safety factor. Our Luer lock design ensures secure attachment to syringes, preventing accidental disconnection during injection procedures. Connection strength testing validates that our needles remain securely attached under normal use forces while allowing intentional disconnection when appropriate.
Core Advantages and Competitive Differentiation
Medical-Grade 304 Stainless Steel – Material Science Excellence
Material selection forms the foundation of needle performance, safety, and reliability. Our choice of medical-grade 304 stainless steel results from comprehensive evaluation of biocompatibility, corrosion resistance, and mechanical properties.
Biocompatibility Performance: 304 stainless steel has undergone extensive biocompatibility testing per ISO 10993 standards, including cytotoxicity, sensitization, and irritation evaluations. The material’s stable chemical composition prevents leaching of harmful substances into animal tissues, while its inert surface minimizes inflammatory responses.
The austenitic crystal structure of 304 stainless steel provides optimal mechanical properties for needle applications. This structure combines adequate hardness for sharpness retention with sufficient toughness to resist fracture under normal use conditions. The material’s work-hardening characteristics actually improve performance during manufacturing, as the drawing process increases surface hardness while maintaining core toughness.
Corrosion Resistance Superiority: The chromium content in 304 stainless steel forms a passive oxide layer that provides exceptional corrosion resistance. This protection remains effective even when exposed to various medications, biological fluids, and cleaning agents commonly encountered in veterinary applications.
The molybdenum content enhances corrosion resistance in chloride-rich environments, important given the prevalence of saline solutions in veterinary medicine. This resistance ensures consistent performance throughout the product’s shelf life and prevents degradation that could affect injection quality or safety.
Mechanical Property Optimization: Our 304 stainless steel specification includes tight control of carbon content to optimize mechanical properties. Lower carbon content improves ductility and corrosion resistance, while precise control ensures consistent performance across production batches.
Heat treatment protocols specific to needle manufacturing enhance the material’s properties. Our proprietary thermal processing creates an optimal balance of hardness and toughness, ensuring sharp needle points that resist deformation while maintaining sufficient flexibility to prevent brittle fracture.
CE Certification – International Quality Assurance Authority
CE certification represents compliance with European Union medical device regulations—among the world’s most stringent quality standards. This certification validates our CE certified veterinary supplies safety, efficacy, and quality across all aspects of design, manufacturing, and performance.
The certification process involves comprehensive technical documentation review, quality management system assessment, and product testing verification. Notified bodies conduct rigorous evaluation of design controls, risk management processes, clinical evaluation data, and manufacturing quality systems.
Technical File Requirements include detailed design specifications, risk analysis documentation, biocompatibility test reports, sterility validation data, and clinical performance studies. This documentation provides complete traceability of design decisions and validates that all regulatory requirements have been met.
Quality Management System Assessment verifies that our ISO 13485 quality management system meets European regulatory requirements. This includes evaluation of design controls, supplier management, production controls, and post-market surveillance systems.
Product Testing Verification confirms that our veterinary disposable needles meet all applicable European standards for medical devices. Testing includes dimensional verification, mechanical property validation, biocompatibility confirmation, and sterility testing according to harmonized European standards.
ISO 13485 Quality Management System – Systematic Excellence Assurance
ISO 13485 represents the global standard for medical device quality management systems, requiring systematic control of all processes affecting product quality and safety. Our implementation goes beyond mere compliance, creating a culture of continuous improvement and quality excellence.
Design Control Implementation: Our design control system ensures systematic development from initial concept through final product launch. Design inputs capture user needs and regulatory requirements, design outputs specify product characteristics, design verification confirms outputs meet inputs, and design validation demonstrates the product meets user needs in actual use conditions.
Risk Management Integration: ISO 14971 risk management principles are embedded throughout our quality system. We systematically identify potential hazards, estimate and evaluate risks, implement risk controls, and monitor risk control effectiveness. This approach ensures patient safety while enabling innovation and performance optimization.
Supplier Management Excellence: Our approved supplier program ensures that all materials and components meet our quality standards. Supplier audits, incoming inspection procedures, and continuous monitoring ensure consistent input quality that supports final product excellence.
Production Process Controls: Statistical process control, equipment qualification, and environmental monitoring ensure consistent manufacturing conditions. Our process validation programs demonstrate that manufacturing processes consistently produce products meeting predetermined specifications.
3. Functional Features and Technical Innovation
Primary Function Modules and Operational Excellence
Precision Injection Capability – Advanced Fluid Dynamics Engineering
Achieving precision in veterinary injection needles requires sophisticated understanding of fluid mechanics, animal anatomy, and injection physics. Our needles incorporate multiple engineering innovations to optimize injection performance across diverse applications.
Needle Tip Geometry Optimization: Our veterinary disposable needles feature precision-engineered elliptical geometry developed through computational modeling and clinical testing. This design minimizes penetration force while reducing tissue trauma—critical for animal comfort and cooperation during procedures.
The tip angle is optimized for different animal skin types. Cattle and horses have thick, tough hide requiring steeper tip angles for effective penetration. Cats and small dogs have thin, delicate skin better served by gentler tip geometries. Our manufacturing process can accommodate these variations within our standard product line.
Penetration Force Minimization: Advanced surface treatments reduce friction during tissue penetration. Our proprietary siliconization process creates an ultra-thin lubricating layer that significantly reduces insertion force while maintaining biocompatibility. This treatment is particularly beneficial for anxious animals or repeated procedures.
Surface roughness control at the nanometer level ensures smooth tissue penetration. We achieve surface finishes of Ra 0.1 micrometers or better through multi-stage polishing processes, creating surfaces that glide smoothly through tissues with minimal drag.
Flow Rate Optimization: Internal diameter specifications are precisely calculated using fluid dynamics principles to optimize flow rates for different medication types. Aqueous solutions including most vaccines and injectable medications flow easily through smaller gauges (20G-25G), enabling comfortable injections with minimal tissue trauma. These solutions generally don’t require large internal diameters for practical flow rates.
Oil-based vaccines and thick suspensions require larger internal diameters (14G-18G) to achieve reasonable injection times. Attempting to use undersized gauges with viscous medications can result in excessive injection pressure, needle blockage, or incomplete medication delivery.
Wall thickness optimization balances flow capacity with structural strength. Our proprietary calculations ensure maximum internal volume while maintaining needle integrity under normal use pressures. This optimization is particularly important for thick medications that generate higher injection pressures.
Blood Collection Functionality – Specialized Hemodynamic Design
Blood collection presents unique challenges requiring specialized needle characteristics. Blood, as a non-Newtonian fluid containing cellular components, behaves differently from liquid medications, necessitating design modifications for optimal collection performance.
Hemolysis Prevention Technology: Our internal surface treatments minimize blood cell damage during collection. Ultra-smooth internal surfaces reduce shear forces that can rupture red blood cells, ensuring sample integrity for accurate laboratory analysis. Surface treatments also prevent platelet activation that could compromise coagulation studies.
Flow Dynamics Optimization: Blood collection requires careful balance between collection speed and sample quality. Our internal diameter calculations account for blood viscosity variations across different animal species and health conditions. Larger animals with higher blood viscosity benefit from slightly larger internal diameters, while smaller animals require more precise flow control.
Collection Efficiency Enhancement: Needle design incorporates features to minimize collection time while maintaining sample quality. This includes optimized internal geometry to reduce turbulence and pressure optimization to maintain steady flow rates without excessive vacuum pressure.
Multi-Angle Adaptability System – Ergonomic Engineering Excellence
Veterinary procedures often require injections at challenging angles and in difficult positions. Our length specifications create a comprehensive adaptability system accommodating diverse operational requirements.
Length Specification Engineering: Each length option results from extensive ergonomic analysis and clinical observation. We studied veterinary injection techniques across multiple practice types to identify optimal length ranges for different applications.
Short lengths (16-25mm) serve subcutaneous injections and surface procedures where precise depth control is critical. These lengths provide excellent tactile feedback, allowing practitioners to feel tissue layers and control injection depth accurately.
Medium lengths (25-38mm) accommodate most intramuscular injections while maintaining good control and flexibility. This range serves the majority of routine veterinary procedures across various animal sizes.
Extended lengths (38-76mm) enable deep tissue access in large animals while maintaining structural integrity. These lengths require enhanced manufacturing precision to prevent flexibility that could compromise injection accuracy.
Structural Strength Engineering: Longer needles face increased bending forces during use, requiring careful material and geometric optimization. Our strength calculations ensure adequate rigidity for controlled injection while preventing brittleness that could lead to breakage.
Finite element analysis validates needle performance under various loading conditions. We simulate injection forces, lateral loads from animal movement, and handling stresses to ensure reliable performance across all expected use scenarios.
Technical Parameters and Performance Indicators
Manufacturing Precision and Quality Control Systems
Dimensional Accuracy Achievement: Our manufacturing processes achieve dimensional tolerances that exceed industry standards, ensuring consistent performance across all production batches.
Wall thickness control at ±0.005mm represents exceptional precision in needle manufacturing. This accuracy requires advanced drawing equipment, precise die maintenance, and continuous monitoring throughout production. Statistical process control ensures that dimensional variation remains well within specified limits.
Outer diameter precision of ±0.01mm ensures consistent penetration characteristics and tissue trauma levels. This precision is achieved through multi-stage drawing processes with intermediate annealing to relieve work hardening stresses.
Length accuracy of ±0.1mm is maintained through laser cutting technology with automated length measurement and feedback control. This precision ensures that practitioners can rely on consistent needle performance across different production lots.
Surface Quality Excellence: Surface finish quality directly impacts injection performance and user experience. Our multi-stage finishing process achieves mirror-like surface quality that reduces friction and improves injection smoothness.
Internal surface roughness below Ra 0.1 micrometers ensures smooth medication flow and easy cleaning of internal surfaces during manufacturing. This smoothness is achieved through specialized honing processes that create consistent surface texture throughout the internal bore.
External surface treatments include cleaning, passivation, and siliconization processes that enhance biocompatibility and reduce insertion force. Each treatment step is validated through surface analysis and performance testing.
Sterility Assurance and Validation Systems
Gamma Irradiation Sterilization Excellence: Our sterilization process uses Cobalt-60 gamma irradiation to achieve the required sterility assurance level. Gamma irradiation provides several advantages over other sterilization methods: complete penetration through packaging and product, no chemical residues, and validation through well-established protocols.
Dose distribution studies ensure uniform sterilization throughout each product unit. We use dosimetry systems to map radiation dose distribution and verify that all product areas receive adequate sterilization dose. This mapping process is repeated for each product configuration to ensure complete sterility assurance.
Biological indicator validation uses Geobacillus stearothermophilus spores—highly resistant to gamma radiation—to verify sterilization effectiveness. These indicators are placed in the most challenging locations within the product load, providing conservative validation of sterilization efficacy.
Package Integrity and Barrier Performance: Our packaging systems provide multiple levels of protection to maintain sterility from sterilization through use. Primary packaging creates an immediate sterile barrier using medical-grade materials validated for gamma irradiation compatibility.
Seal strength testing ensures package integrity under normal handling and storage conditions. We conduct accelerated aging studies to verify that package seals maintain integrity throughout the product’s shelf life. Dye penetration testing validates seal quality and identifies any potential leak paths.
Shelf Life Validation Through Comprehensive Stability Studies: Our 5-year shelf life designation results from extensive stability testing programs that exceed regulatory requirements for safety margin.
Real-time stability studies monitor product performance under recommended storage conditions (25°C ± 2°C, 60% ± 5% RH) for the full shelf life period. Testing includes dimensional stability, mechanical properties, package integrity, and sterility maintenance.
Accelerated stability studies at elevated temperature and humidity conditions (40°C ± 2°C, 75% ± 5% RH) provide predictive data for long-term stability. These studies follow Arrhenius kinetics to model degradation rates and predict performance at normal storage conditions.
4. User Experience and Ergonomic Design
Ease of Use and Operational Efficiency
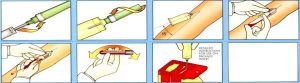
Package Opening Experience – Human Factors Engineering
The seemingly simple act of opening a needle package represents a critical user interaction that can impact procedural efficiency and safety. Our package design incorporates human factors engineering principles to optimize this experience.
Single-Hand Operation Capability: Veterinary procedures often require one hand to restrain the animal while the other manages equipment. Our tear-strip design enables secure single-handed package opening without compromising sterility or safety.
The tear strip location and size result from extensive user testing with veterinary professionals. The strip is positioned for natural thumb and finger placement, with sufficient size for secure grip even when wearing gloves. The tear force is calibrated to provide positive opening feedback while preventing accidental opening during normal handling.
Sterility Maintenance During Opening: Package design maintains sterility throughout the opening process. The tear pattern creates a controlled opening that doesn’t compromise the sterile internal environment. Opening technique training emphasizes maintaining sterility while achieving efficient access to the needle.
Penetration Performance – Biomechanical Optimization
Smooth, controlled tissue penetration enhances both animal comfort and procedural success. Our veterinary disposable needles design incorporates multiple features to optimize penetration performance across diverse animal types and conditions.
Sharpness Retention Technology: Needle tip geometry is designed for sustained sharpness throughout the penetration process. Our proprietary tip design maintains cutting efficiency as it progresses through different tissue layers, from tough outer skin to softer subcutaneous tissue.
Manufacturing processes include specialized tip finishing that creates consistently sharp points with minimal tip defects. Quality control includes automated sharpness testing that measures penetration force under standardized conditions.
Tissue Trauma Minimization: Beyond sharpness, our design minimizes tissue trauma through careful consideration of needle shaft geometry and surface treatments. The transition from needle tip to shaft is optimized to reduce tissue tearing as the needle advances.
Surface treatments reduce friction between needle and tissue, allowing smooth advancement with minimal force. This reduced friction translates to less tissue disruption and faster healing at injection sites.
Interface Design and User Interaction Excellence
Visual Recognition Systems – Cognitive Engineering Application
Rapid, accurate needle selection is critical in busy veterinary environments where wrong gauge selection could impact treatment outcomes or animal safety. Our color-coding system applies cognitive engineering principles to optimize recognition speed and accuracy.
Color Psychology Integration: Color selection incorporates both practical visibility requirements and psychological associations. Larger gauges use colors psychologically associated with strength and reliability (deep blues, strong greens), while smaller gauges use colors suggesting precision and delicacy (lighter blues, subtle purples).
Color contrast optimization ensures clear differentiation under various lighting conditions common in veterinary facilities. Our color palette maintains effectiveness under fluorescent, LED, and natural lighting while accommodating color vision deficiencies that affect approximately 8% of male users.
Information Hierarchy Design: Package labeling follows information hierarchy principles that prioritize critical identification data. Gauge size receives primary visual emphasis through large, bold typography and high-contrast color application.
Secondary information (length, lot number, expiration date) uses smaller typography and lower contrast positioning that doesn’t interfere with primary identification but remains easily accessible when needed. This hierarchical approach reduces cognitive load during high-stress procedures.
Tactile Feedback Systems – Sensory Design Integration
Injection procedures rely heavily on tactile feedback to guide needle placement and injection depth. Our design optimizes tactile feedback to enhance procedural accuracy and safety.
Penetration Feedback Enhancement: Needle design provides distinct tactile feedback as it progresses through different tissue layers. The resistance change when penetrating from skin to subcutaneous tissue provides clear indication of successful penetration depth.
Different tissue types provide characteristic resistance patterns that experienced practitioners learn to interpret. Our needle design enhances these natural feedback mechanisms without creating artificial resistance that could impede smooth injection.
Depth Control Optimization: Length specifications provide tactile reference points for injection depth control. Practitioners develop tactile memory for appropriate insertion depths with specific needle lengths, improving injection accuracy and consistency.
Hub design provides tactile reference points that help practitioners maintain consistent grip and control throughout injection procedures. Surface texturing and dimensional optimization create positive tactile feedback without compromising comfort during extended use.
5. Commercial Information and Business Excellence
Pricing Strategy and Package Options – Value-Based Market Positioning
Total Cost of Ownership Analysis
Our pricing strategy reflects comprehensive Total Cost of Ownership (TCO) analysis that demonstrates superior value despite higher initial unit costs compared to reusable alternatives. This analysis incorporates direct costs, indirect costs, and risk-adjusted costs to provide accurate economic comparisons.
Direct Cost Components include purchase price, shipping costs, and storage costs. While our per-unit cost exceeds reusable needle costs, the elimination of reprocessing expenses (cleaning, sterilization, validation) often results in lower total direct costs for most applications.
Indirect Cost Benefits include time savings from eliminated reprocessing, reduced labor costs, improved efficiency from consistent performance, and enhanced procedural predictability. These benefits often exceed direct cost differences, particularly in high-volume veterinary applications.
Risk-Adjusted Cost Analysis incorporates potential costs of infection events, needle failure, and regulatory non-compliance. The elimination of cross-contamination risks provides substantial risk-adjusted value, particularly for practices serving high-value animals or operating under strict regulatory oversight.
Tiered Packaging Strategy – Customer-Centric Solutions
Our packaging options address diverse customer needs across different practice types and operational scales, from individual practitioners to large commercial operations.
Individual Unit Packaging serves specialty applications, trial usage, and practices with diverse needs requiring multiple gauge options in small quantities. This packaging provides maximum flexibility while minimizing inventory investment.
Unit packaging incorporates premium materials and enhanced protection to justify higher per-unit costs. Each unit receives individual inspection and quality verification, ensuring premium performance for demanding applications.
Standard Box Packaging (100 units) represents our volume-optimized solution for typical veterinary practices. This packaging size balances inventory management convenience with reasonable unit costs, serving the majority of our customer base.
Box packaging utilizes efficient protective materials while maintaining individual unit sterility. Secondary packaging provides protection during shipping and storage while enabling efficient inventory management and dispensing.
Bulk Packaging (1,000 units) serves high-volume operations requiring maximum cost efficiency. Large farms, research facilities, and high-volume practices benefit from reduced per-unit costs and simplified inventory management.
Bulk packaging incorporates institutional-grade protection and handling features designed for high-throughput use environments. Package design enables efficient dispensing while maintaining individual unit protection.
Mixed Gauge Kits provide comprehensive solutions for practices requiring multiple gauge options. These kits eliminate individual gauge inventory management while ensuring appropriate gauge availability for diverse procedures.
Kit composition reflects typical usage patterns observed across different practice types. Mixed ratios optimize inventory turnover while minimizing stock-out risks for any individual gauge size.
Distribution Channels and Sales Excellence
Authorized Dealer Network – Strategic Market Coverage
Our national authorized dealer network provides comprehensive market coverage while maintaining consistent service quality and technical support capabilities across all regions.
Dealer Selection Criteria ensure partner quality and capability alignment with our brand standards. Requirements include medical device distribution licensing, professional sales and technical teams, appropriate warehousing and logistics capabilities, and established veterinary industry relationships.
Each dealer undergoes comprehensive evaluation including financial stability assessment, facility inspection, personnel qualification verification, and customer reference validation. This thorough selection process ensures partners capable of representing our brand excellently.
Territory Protection Policies provide dealers with stable business environments and reasonable profit expectations. Exclusive territorial rights prevent destructive price competition while ensuring adequate market coverage and customer service quality.
Training and Support Systems ensure dealer success through comprehensive product knowledge, sales technique training, and technical support capability development. Regular training programs keep dealer personnel current with product innovations and industry developments.
B2B E-Commerce Platforms – Digital Sales Innovation
Digital sales channels provide customers with convenient, efficient procurement options while maintaining professional service standards appropriate for animal healthcare medical devices sales.
Platform Strategy Implementation: We maintain official presence on major B2B platforms including Alibaba, Amazon Business, and specialized medical device marketplaces. Each platform features comprehensive product information, competitive pricing, and streamlined ordering processes.
Platform-specific optimization ensures maximum visibility and conversion rates. Product listings incorporate keyword optimization, detailed specifications, customer reviews, and educational content that supports informed purchasing decisions.
Digital Customer Service Excellence: Online platforms feature integrated customer service including live chat support, technical consultation, and order tracking capabilities. Customers receive the same professional service online as through traditional channels.
Direct Sales Program – Enterprise Customer Focus
Large-volume customers receive dedicated direct sales support providing enhanced service levels and customized solutions appropriate for their scale and complexity.
Enterprise Customer Qualification: Direct sales program serves customers with annual volumes exceeding 100,000 units, including large veterinary hospital groups, major agricultural operations, government procurement programs, and research institutions.
These customers receive dedicated account management, customized pricing structures, priority allocation during supply constraints, and enhanced technical support services including on-site consultation and training.
Customization Capabilities: Direct sales customers can access product customization options including special packaging, private labeling, modified specifications, and custom kit configurations. These services enable customers to optimize their operations while maintaining our quality standards.
International Trade and Global Market Development
Export Market Analysis: Primary export markets include Southeast Asia, Middle East, Africa, and Latin America—regions with growing veterinary sectors and increasing demand for quality animal healthcare medical devices.
Export success factors include competitive pricing relative to European alternatives, comprehensive certification portfolio, and superior customer service compared to traditional Chinese exporters. Our quality-first approach differentiates us in international markets increasingly focused on product safety and reliability.
Regulatory Compliance Portfolio: International sales require extensive regulatory compliance across multiple jurisdictions. Our certification portfolio includes CE marking for European markets, ISO 13485 for global quality recognition, and preparation for FDA registration to access North American markets.
OEM Service Capabilities: Complete OEM manufacturing capabilities serve international partners requiring private label or customized solutions. Services include product specification modification, custom packaging design, regulatory support for local registration, and technical training for distributor personnel.
After-Sales Service and Technical Support Excellence
Comprehensive Quality Guarantee Program
Our quality guarantee program extends beyond basic warranty coverage to provide comprehensive customer protection and confidence in product performance.
Quality Issue Resolution Protocols: All quality concerns receive immediate attention through our structured investigation process. Customer reports trigger automatic quality investigation including lot review, manufacturing record analysis, and potential field testing to identify root causes.
Resolution timelines are clearly defined: standard issues receive response within 48 hours, urgent issues within 24 hours, and emergency situations receive immediate attention. Resolution may include product replacement, credit adjustments, or enhanced technical support depending on investigation findings.
Traceability System Implementation: Complete lot traceability enables rapid identification of affected products and implementation of corrective actions when necessary. Our traceability system links each production lot to specific raw materials, manufacturing conditions, test results, and distribution records.
This system enables precise corrective action scope definition, minimizing customer